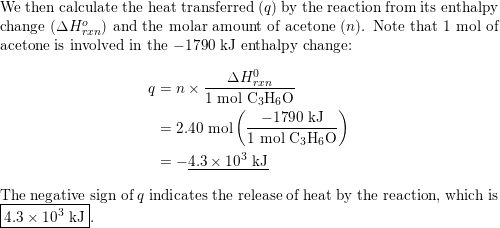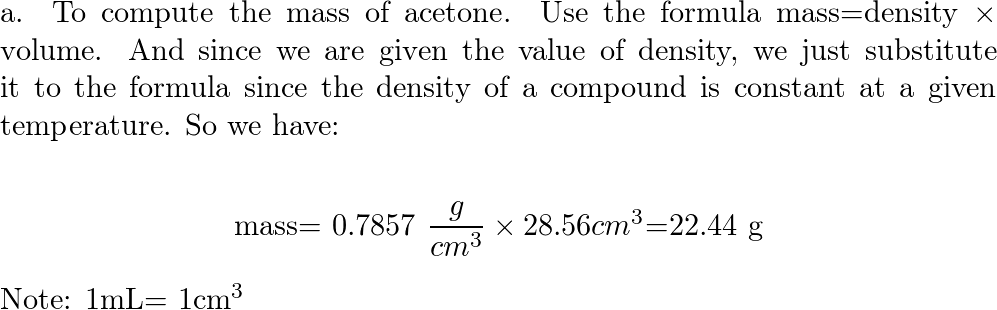Acetone, the solvent in nail polish remover, has a density of 0791 g/mL What is the volume, in mil - YouTube

On mixing 15.0 ml of ethyl alcohol of density 0.792 g ml^-3 with 15 ml of pure water at 4^o C, the resulting solution is found to have a density of 0.924

Mix 4.0 mL of benzaldehyde with 1 mL of acetone in 26 mL of 10% sodium hydroxide solution. (Density of benzaldehyde is 1.04 g/mL, density of acetone is 0.79 g/mL and density

OneClass: The solute is acetone. The density of acetone is .791g/ml. The solvent is acetic acid. The ...

A 1.000 mL sample of acetone, a common solvent used as a paint remover, was placed in a small bottle whose mass was known to be 38.0015 g. The following values were