
Effect of 2-Substituents on Allyl-Supported Precatalysts for the Suzuki–Miyaura Reaction: Relating Catalytic Efficiency to the Stability of Palladium(I) Bridging Allyl Dimers | Organometallics
![Comparative reactivity of allylic alcohols. Conditions: [Pd] ) 0.0055 M... | Download Scientific Diagram Comparative reactivity of allylic alcohols. Conditions: [Pd] ) 0.0055 M... | Download Scientific Diagram](https://www.researchgate.net/publication/244460612/figure/fig1/AS:298491034259456@1448177216307/Comparative-reactivity-of-allylic-alcohols-Conditions-Pd-00055-M-in-water-at-25.png)
Comparative reactivity of allylic alcohols. Conditions: [Pd] ) 0.0055 M... | Download Scientific Diagram

Palladium-Catalyzed, Site-Selective Direct Allylation of Aryl C–H Bonds by Silver-Mediated C–H Activation: A Synthetic and Mechanistic Investigation | Journal of the American Chemical Society

PDF) Palladium complexes with a tridentate PNO ligand. Synthesis of ??1- allyl complexes and cross-coupling reactions promoted by boron compounds

Palladium(II)-Catalyzed Regioselective Hydroesterification of 1,3-Conjugated Enynes with Aryl Formates | Organic Letters

Synergistic Organoboron/Palladium Catalysis for Regioselective N-Allylations of Azoles with Allylic Alcohols | Organic Letters

Palladium‐Catalyzed Electrophilic Allylation Reactions via Bis(allyl) palladium Complexes and Related Intermediates - Szabó - 2004 - Chemistry – A European Journal - Wiley Online Library

Palladium-catalyzed reaction of γ-silylated allyl acetates proceeding through 1,2-shift of a substituent on silicon - ScienceDirect

Palladium‐Catalyzed Electrophilic Allylation Reactions via Bis(allyl) palladium Complexes and Related Intermediates - Szabó - 2004 - Chemistry – A European Journal - Wiley Online Library

Palladium‐Catalyzed Electrophilic Allylation Reactions via Bis(allyl) palladium Complexes and Related Intermediates - Szabó - 2004 - Chemistry – A European Journal - Wiley Online Library

Palladium-catalyzed reaction of γ-silylated allyl acetates proceeding through 1,2-shift of a substituent on silicon - ScienceDirect

Generating Active “L-Pd(0)” via Neutral or Cationic π-Allylpalladium Complexes Featuring Biaryl/Bipyrazolylphosphines: Synthetic, Mechanistic, and Structure–Activity Studies in Challenging Cross-Coupling Reactions | The Journal of Organic Chemistry
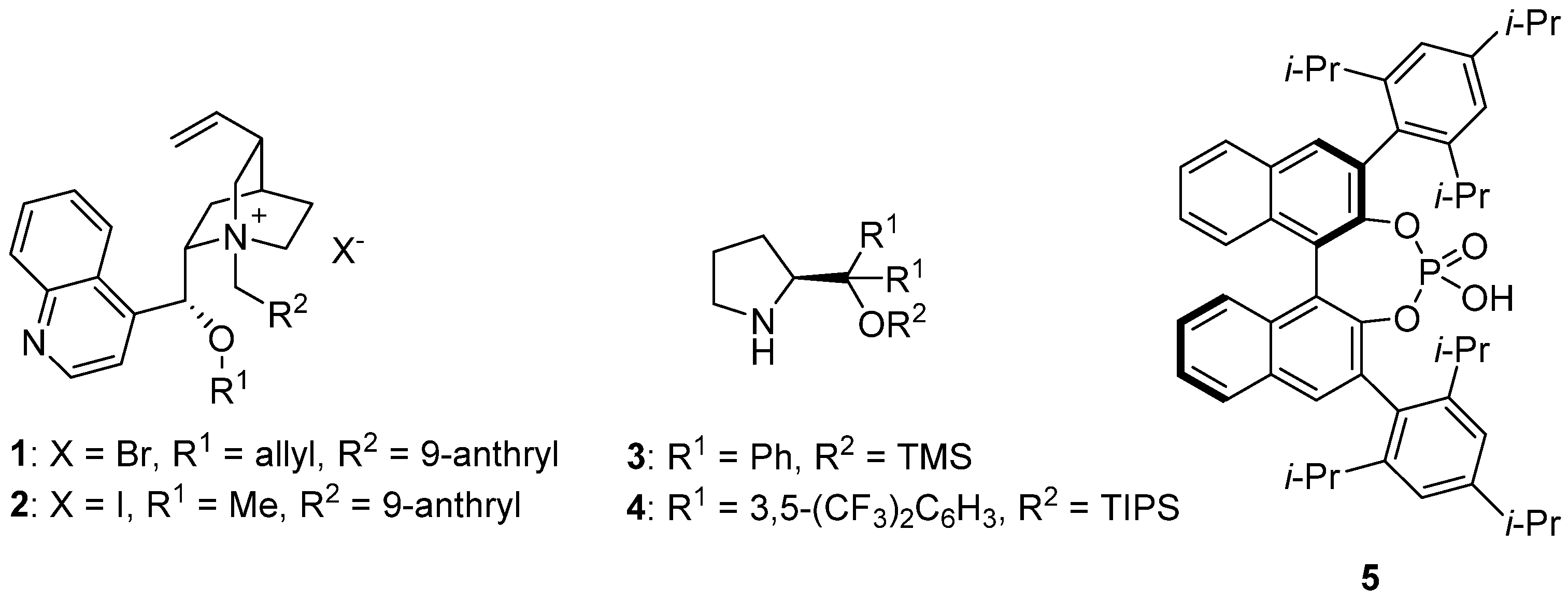
Molecules | Free Full-Text | Palladium and Organocatalysis: An Excellent Recipe for Asymmetric Synthesis

Catalysts | Free Full-Text | Synthesis, Characterization, Solution Behavior and Theoretical Studies of Pd(II) Allyl Complexes with 2-Phenyl-3H-indoles as Ligands

Scheme 3. Part of the mechanism for the palladium catalyzed allylic... | Download Scientific Diagram
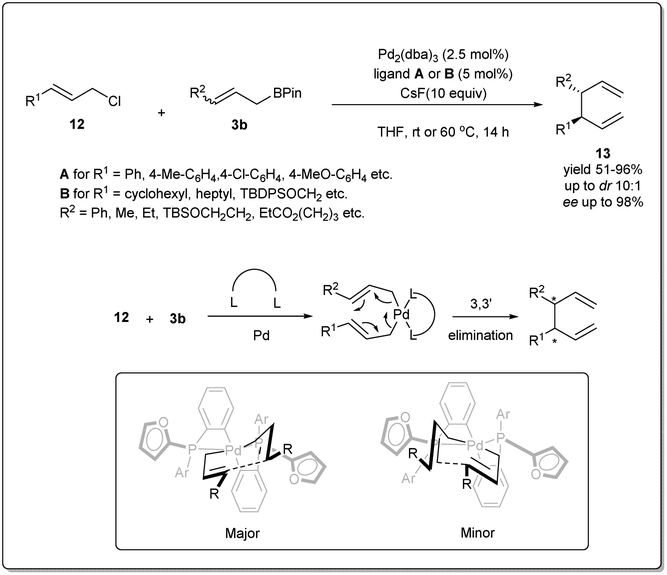
Recent development of allyl–allyl cross-coupling and its application in natural product synthesis - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D2QO01078J
![PDF) Heterolytic Splitting of Allylic Alcohols with Palladium(0)−TPPTS in Water. Stabilities of the Allylphosphonium Salt of TPPTS and of the Ionic Complex [Pd(η 3 -allyl)(TPPTS) 2 ] + PDF) Heterolytic Splitting of Allylic Alcohols with Palladium(0)−TPPTS in Water. Stabilities of the Allylphosphonium Salt of TPPTS and of the Ionic Complex [Pd(η 3 -allyl)(TPPTS) 2 ] +](https://www.researchgate.net/profile/Iyad-Karame/publication/244460612/figure/fig6/AS:671187042566145@1537034869190/Allyl-alcohol-reaction-without-acid-addition-pH-versus-time-Conditions-Pd-00055-M_Q320.jpg)
PDF) Heterolytic Splitting of Allylic Alcohols with Palladium(0)−TPPTS in Water. Stabilities of the Allylphosphonium Salt of TPPTS and of the Ionic Complex [Pd(η 3 -allyl)(TPPTS) 2 ] +
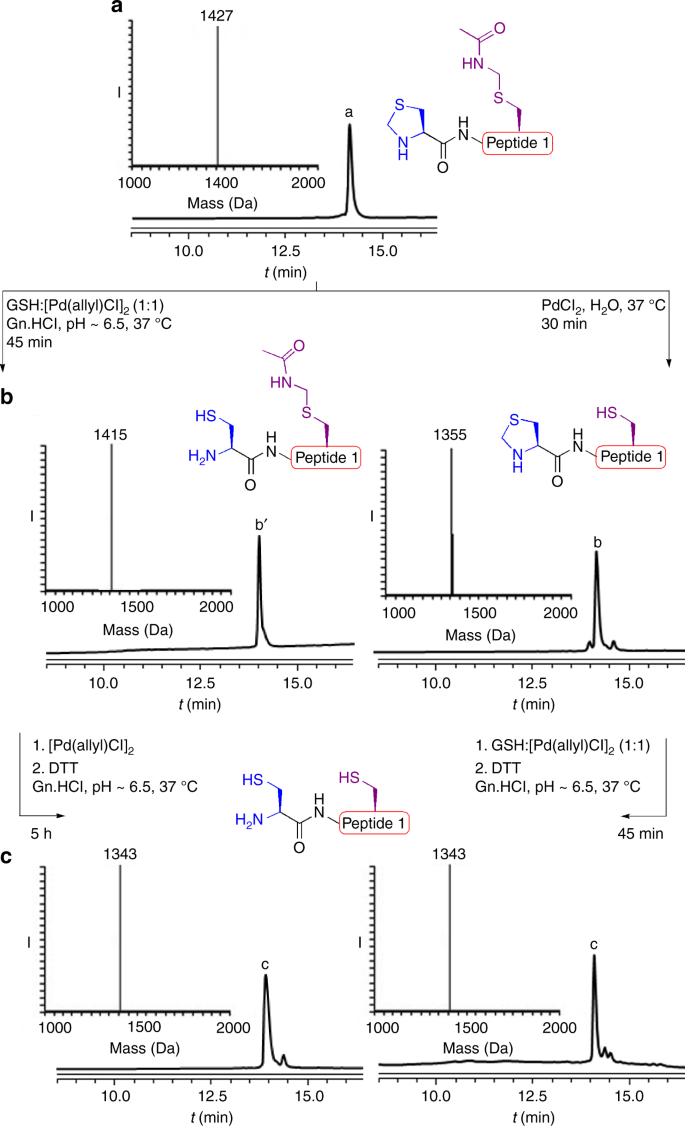
Palladium prompted on-demand cysteine chemistry for the synthesis of challenging and uniquely modified proteins | Nature Communications

Asymmetric allylic substitution by chiral palladium catalysts: Which is more reactive, major π-allyl Pd(II) species or minor π-allyl species? - ScienceDirect
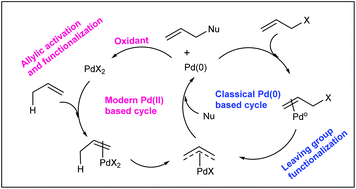
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)

Palladium-catalyzed reaction of γ-silylated allyl acetates proceeding through 1,2-shift of a substituent on silicon - ScienceDirect

Palladium‐Catalyzed Allyl Cross‐Coupling Reactions with In Situ Generated Organoindium Reagents - Lee - 2011 - Chemistry – An Asian Journal - Wiley Online Library
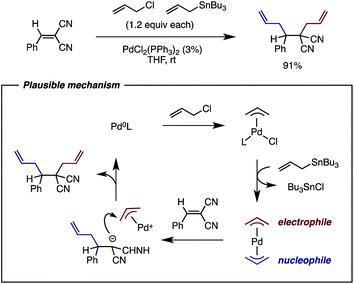
Palladium -catalyzed tetraallylation of C 60 with allyl chloride and allylstannane : mechanism, regioselectivity, and enantioselectivity - Chemical Science (RSC Publishing) DOI:10.1039/C2SC21126B


