Freezing Point Depression When the rate of freezing is the same as the rate of melting, the amount of ice and the amount of water won't change. The. - ppt download
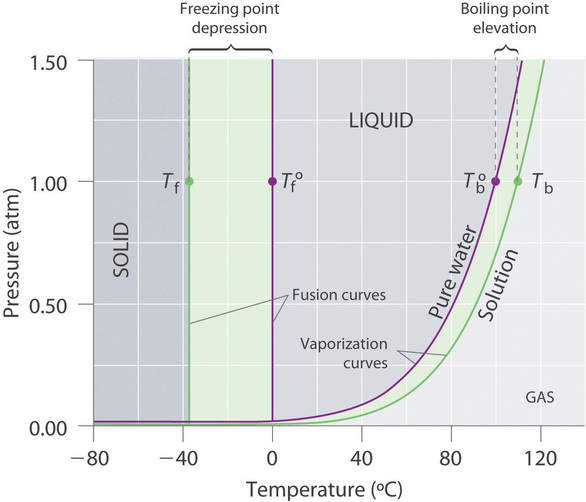
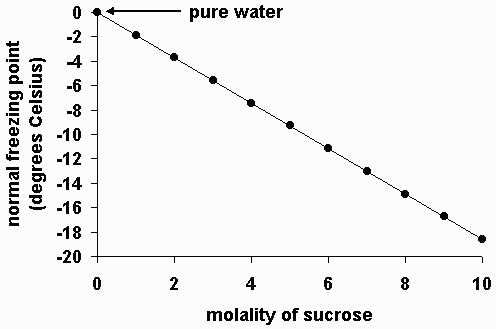
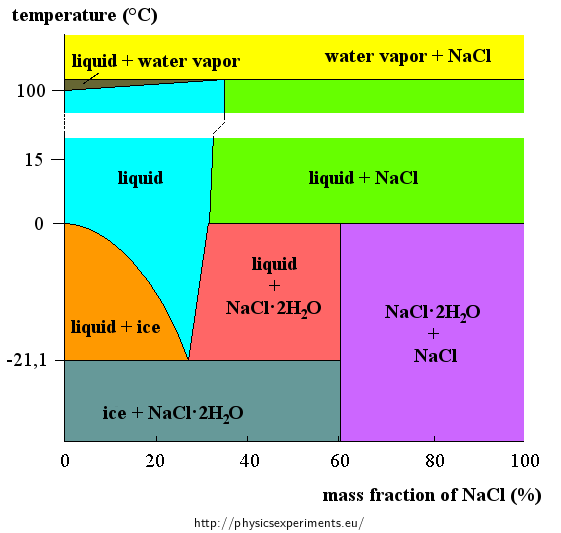
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts

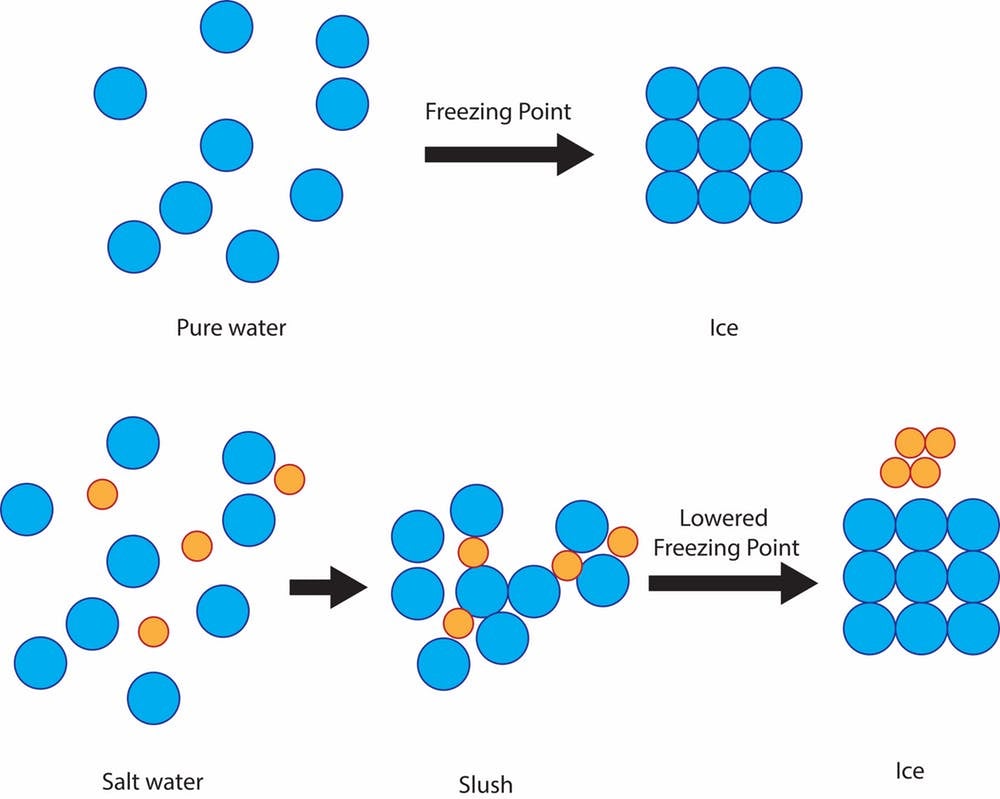
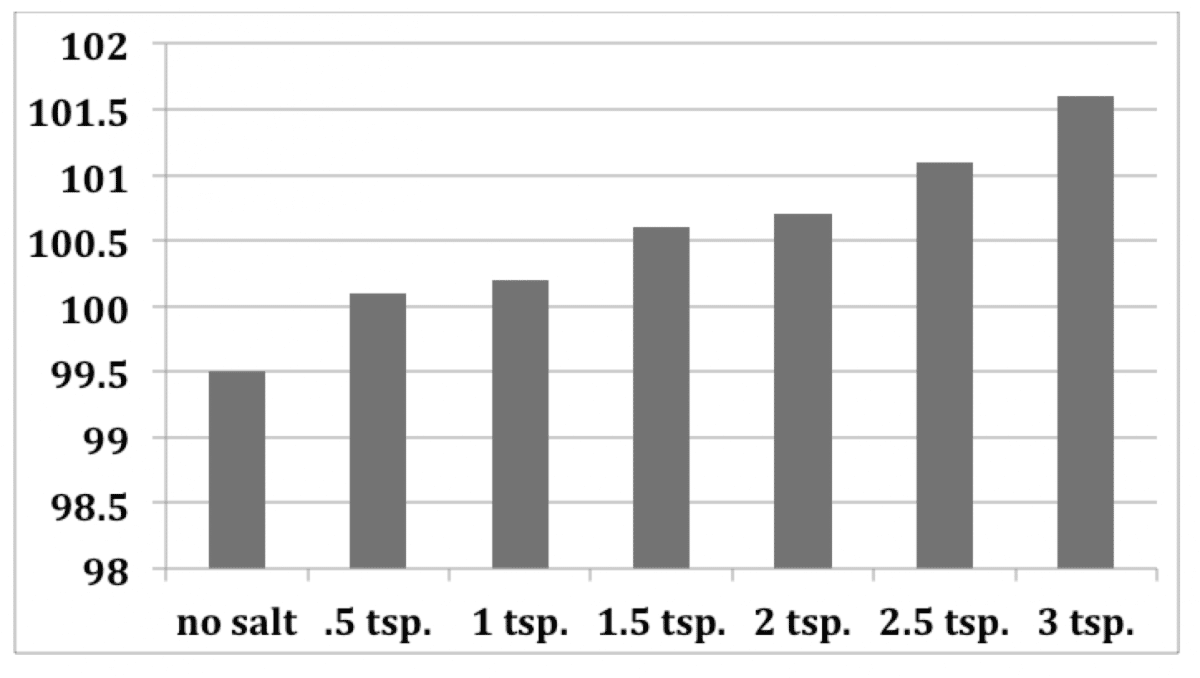
Changing melting and boiling points. Putting salt on sidewalks and roads in the winter helps because it lowers the freezing point of water by a few degrees. - ppt download






:max_bytes(150000):strip_icc()/water-in-steel-pan-with-herbs-and-salt-being-added-making-brine-145063802-57a770ea3df78cf459166075.jpg)