Maocao Hoom 27.56-in W 40000-BTU Brown Portable Magnesium Oxide Propane Gas Fire Pit in the Gas Fire Pits department at Lowes.com
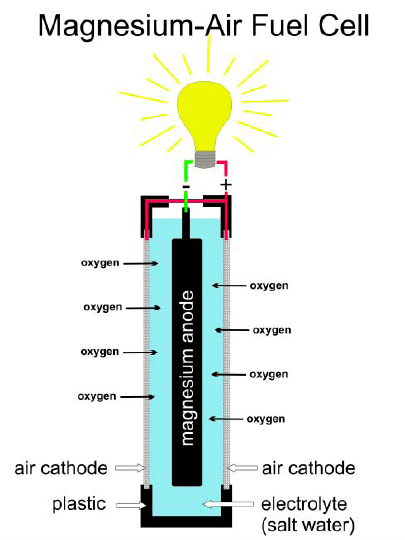
Ecore Global & MagPower, Canada Will Introduce a prototype of Magnesium/Air Fuel Cell at WCTA Breakfast – CleanTech Alliance

Magnesium powder burning in gas jar - an exothermic reaction - emitting a bright light. Science experiment in a laboratory setting. See A2021A, A20219 Stock Photo - Alamy

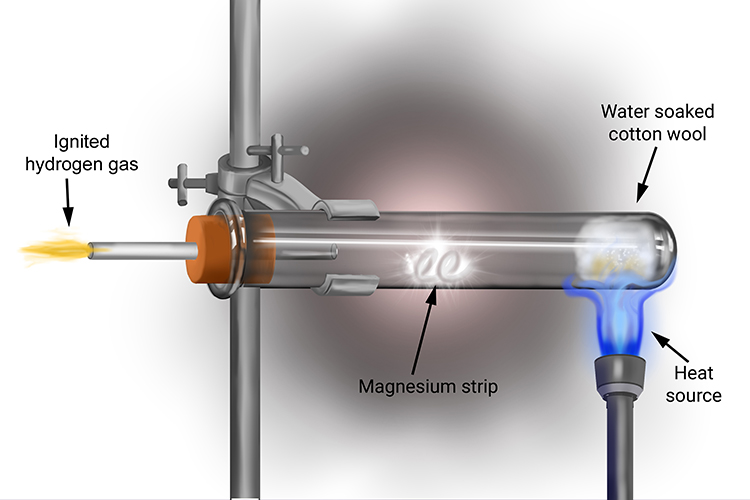
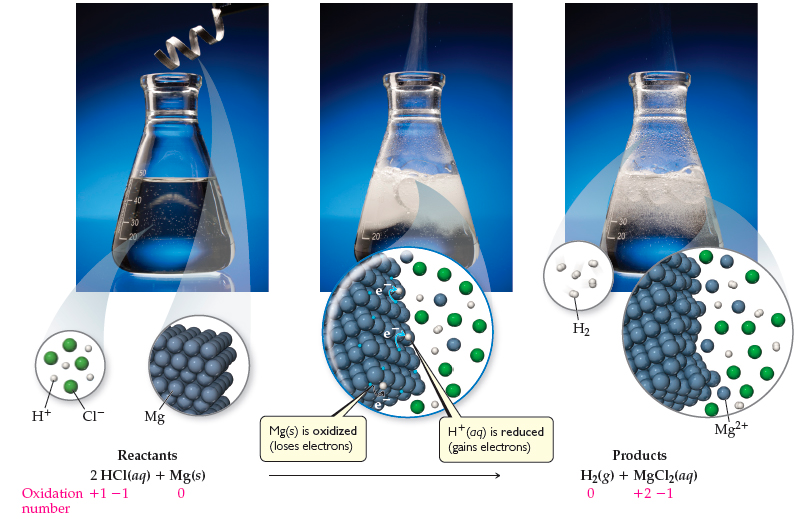
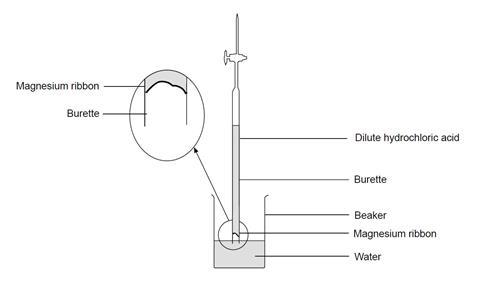
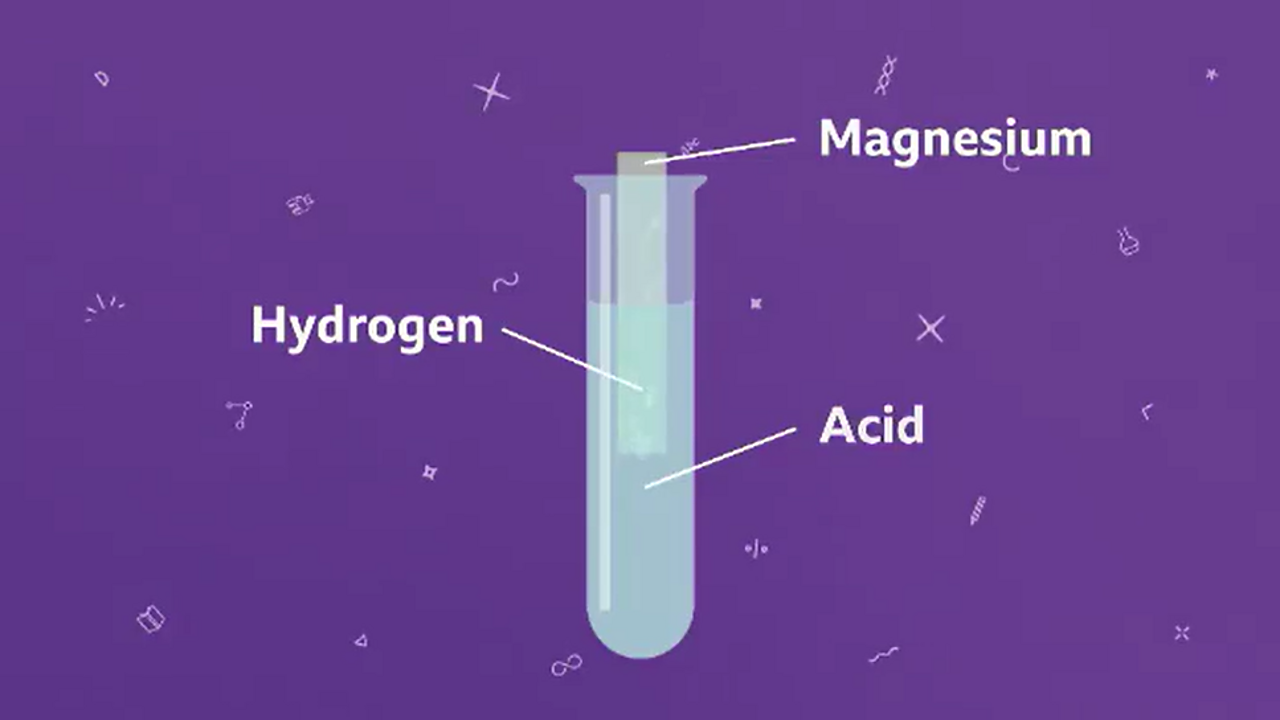
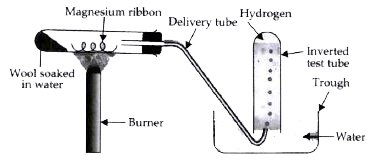
Question Video: Identifying the Name of the Gas Produced When Magnesium Metal Reacts with Hydrochloric Acid | Nagwa

Development of a Model System for Gas Cavity Formation Behavior of Magnesium Alloy Implantation | ACS Biomaterials Science & Engineering

Magnesium carbonate reacting with hydrochloric acid. This reaction produces bubbles of carbon dioxide gas, as well as magnesium chloride and water Stock Photo - Alamy

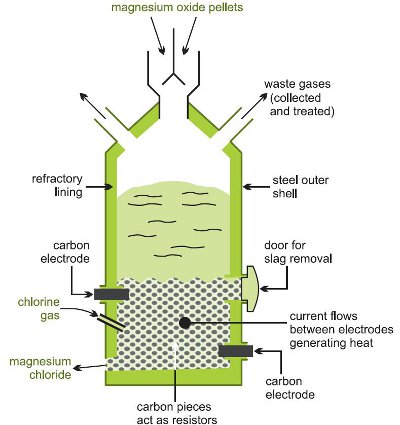
Magnesium Elektron Introduces Proprietary SoluMag™ Alloy for Making Down-Hole Oil and Gas Industry Tools That Dissolve After Use | Business Wire

Amazon.com: Natural Magnesium Water - 1000mg Magnesium in 1000ml ''Donat Mg'' Bulk Pack 6x1L : Health & Household
%20ribbon%20in%20a%20beaker%20reacting%20with%20hydrochloric%20acid%20(HCl).%20This%20reaction%20creates%20magnesium%20chloride%20(MgCl2)%20and%20hydrogen%20gas%20(H2).%20Mg%20%2B%202HCl%20---%20MgCl2%20%2B%20H2.jpg)
Bildagentur | mauritius images | Magnesium (Mg) ribbon in a beaker reacting with hydrochloric acid (HCl). This reaction creates magnesium chloride (MgCl2) and hydrogen gas (H2). Mg + 2HCl --> MgCl2 + H2