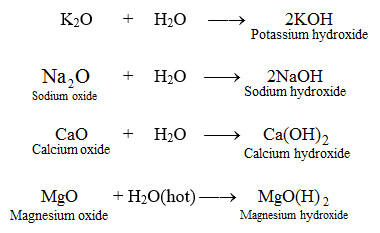Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink
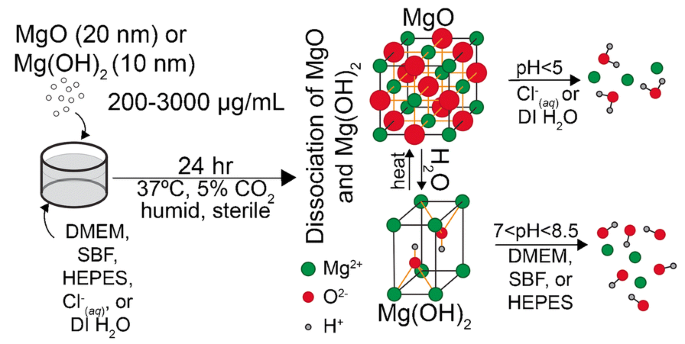
Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink

On some regularities of magnesium oxide solubility in melts with different content of alkaline earth metal chlorides - ScienceDirect

A mixture consists of four substances.The properties of each of these substances are shown in the table.Magnesium nitrate decomposes on heating to form solid magnesium oxide oxygen gas and nitrogen dioxide gas.

SOLVED:Discuss the interpretation of the observation that magnesium oxide is more soluble in aqueous magnesium chloride than in pure water,

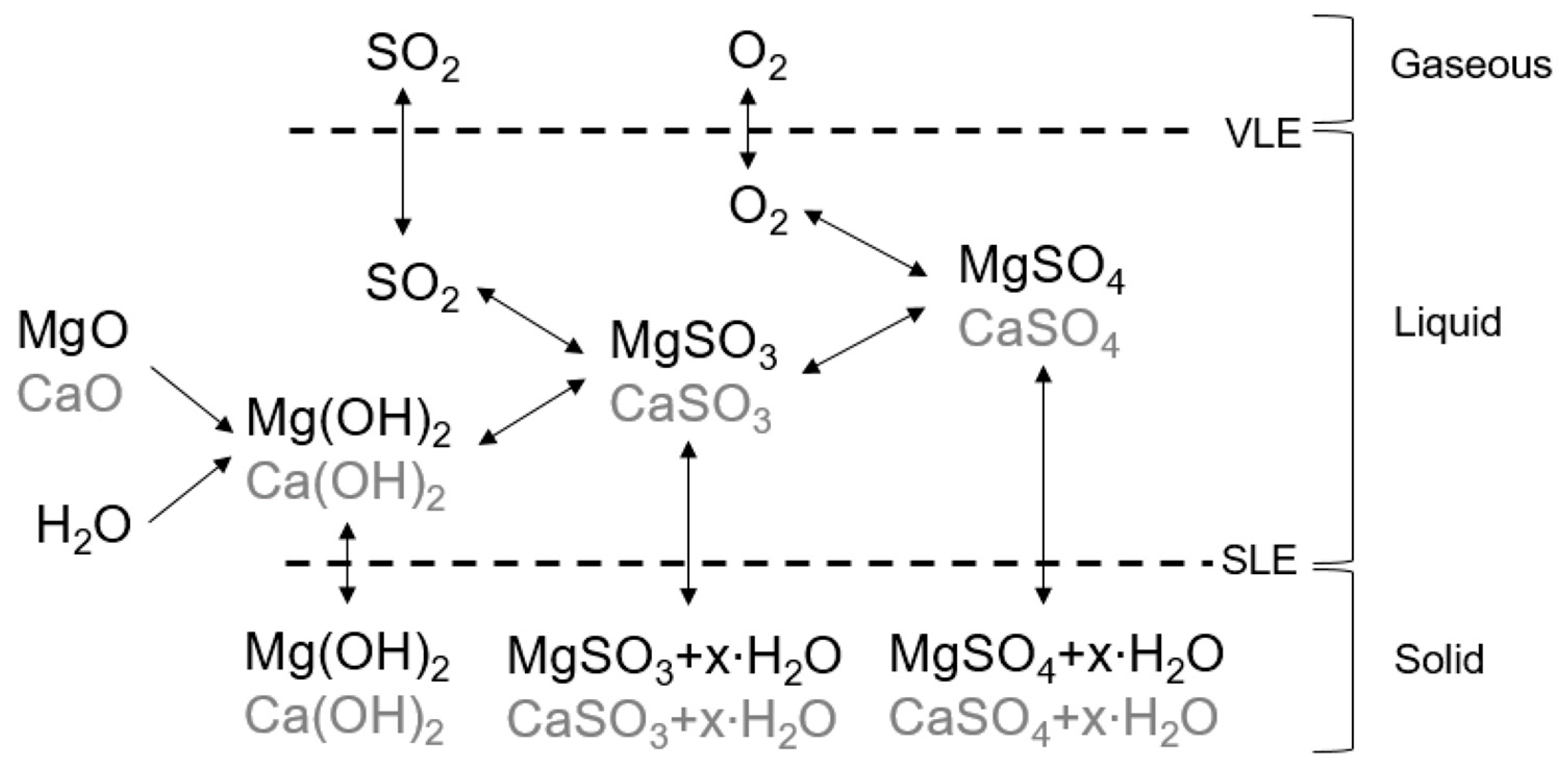
The solubility of magnesium sulphite hydrates calculated by Aspen Plus®... | Download Scientific Diagram