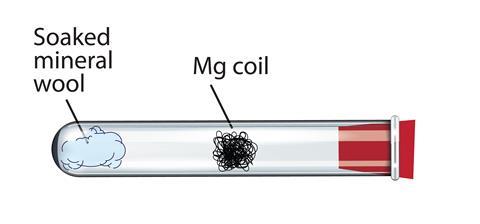
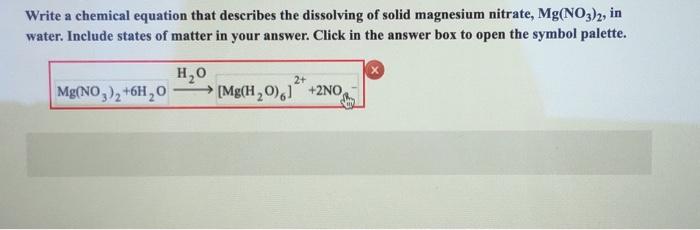
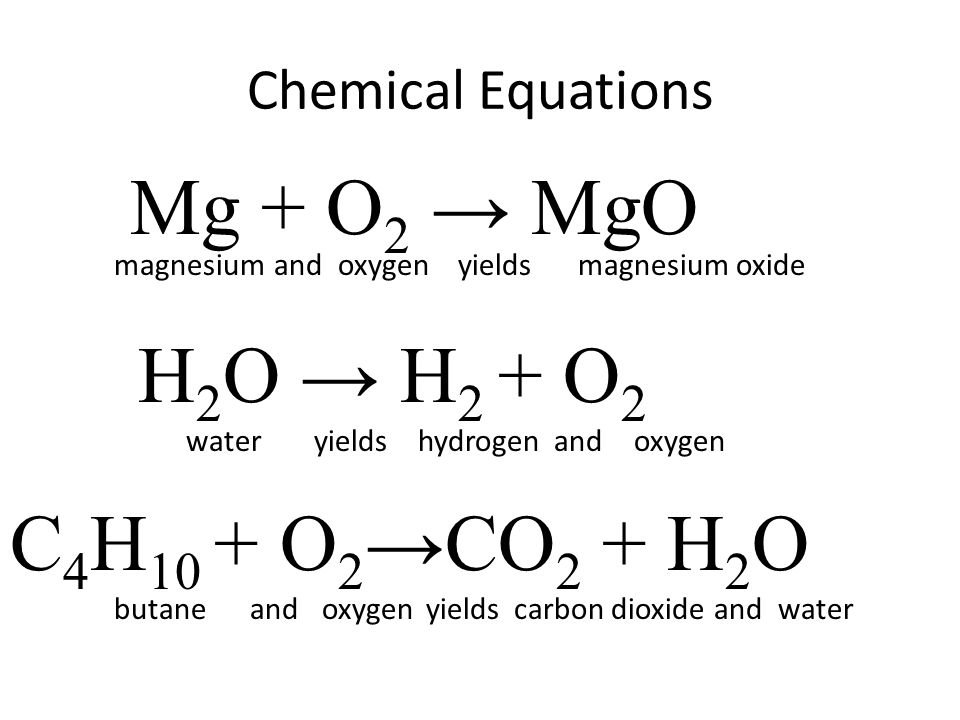
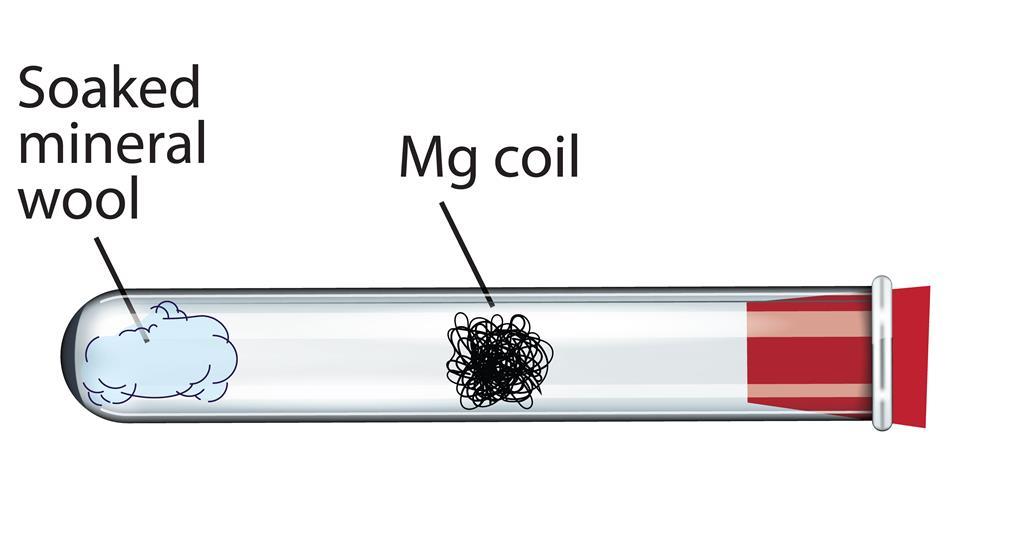
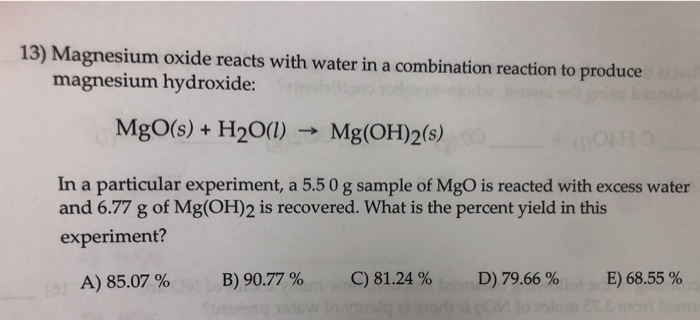What happens when magnesium oxide is dissolved in water ? Write a word equation for this process. Name the type of change which takes place.

why calcium and magnesium floats on water - Science - Metals and Non-metals - 13061069 | Meritnation.com

SOLVED: Magnesium metal reacts with water via the following balanced chemical equation: Mg (s) + 2 HzO Mg(OH)z (s) + Hz (g) If 4.73 g of magnesium is allowed to react with