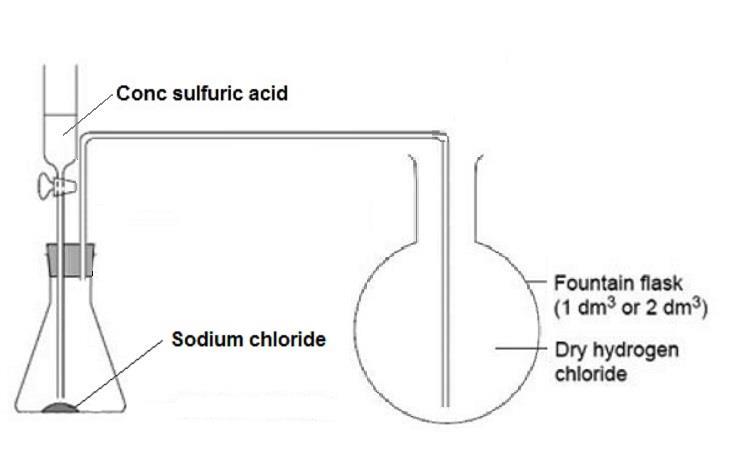
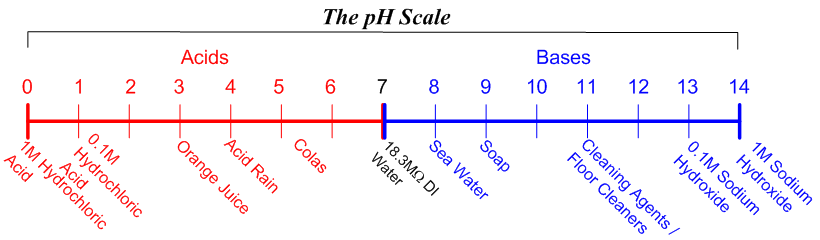
Neutralizing Solutions with Sodium Hydroxide | Process & Chemical Formula - Video & Lesson Transcript | Study.com

Name the products formed in each case when: a. hydrochloric acid reacts with caustic soda b.granulated zinc - Brainly.in

Sulfuric Acid, Hydrochloric Acid, Nitric Acid, Glacial Acetic Acid, Formaldehyde, Formic Acid Supplier
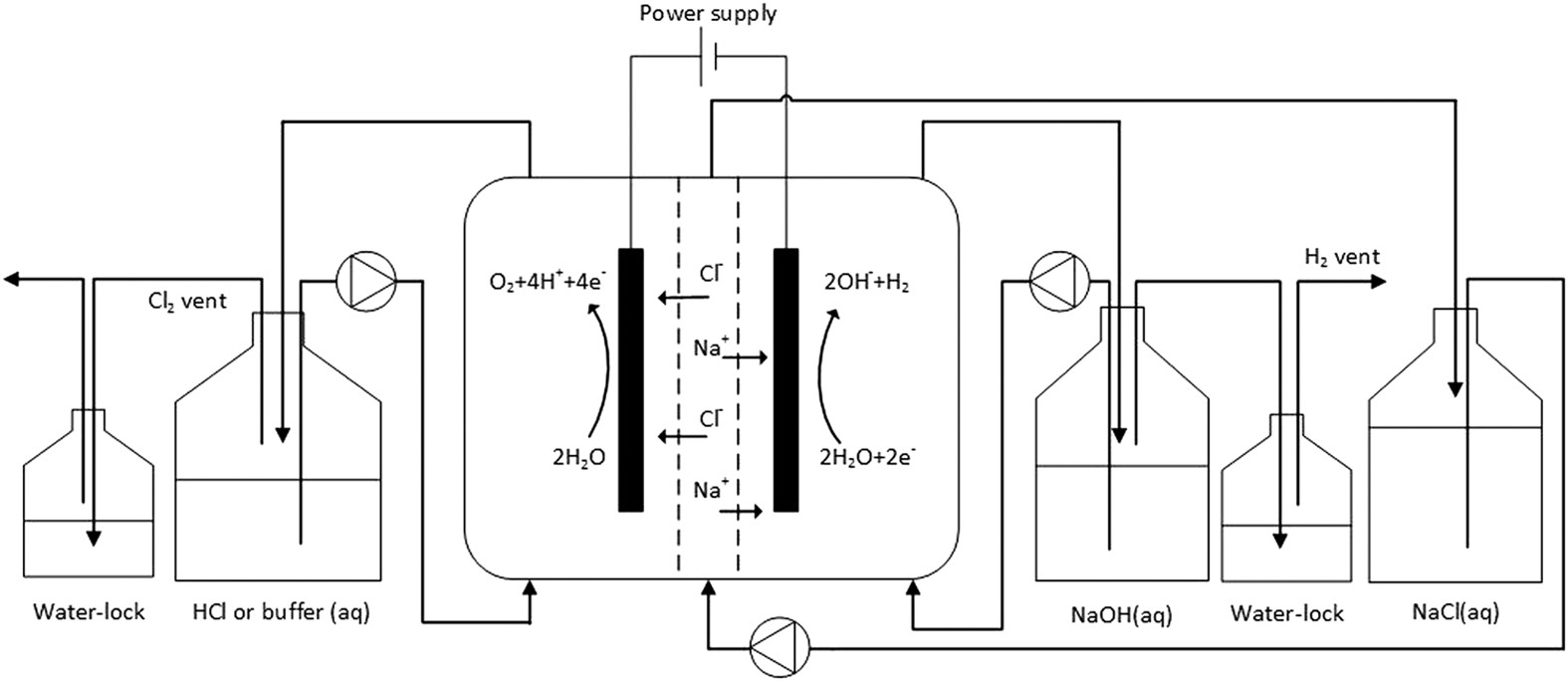
Direct anodic hydrochloric acid and cathodic caustic production during water electrolysis | Scientific Reports
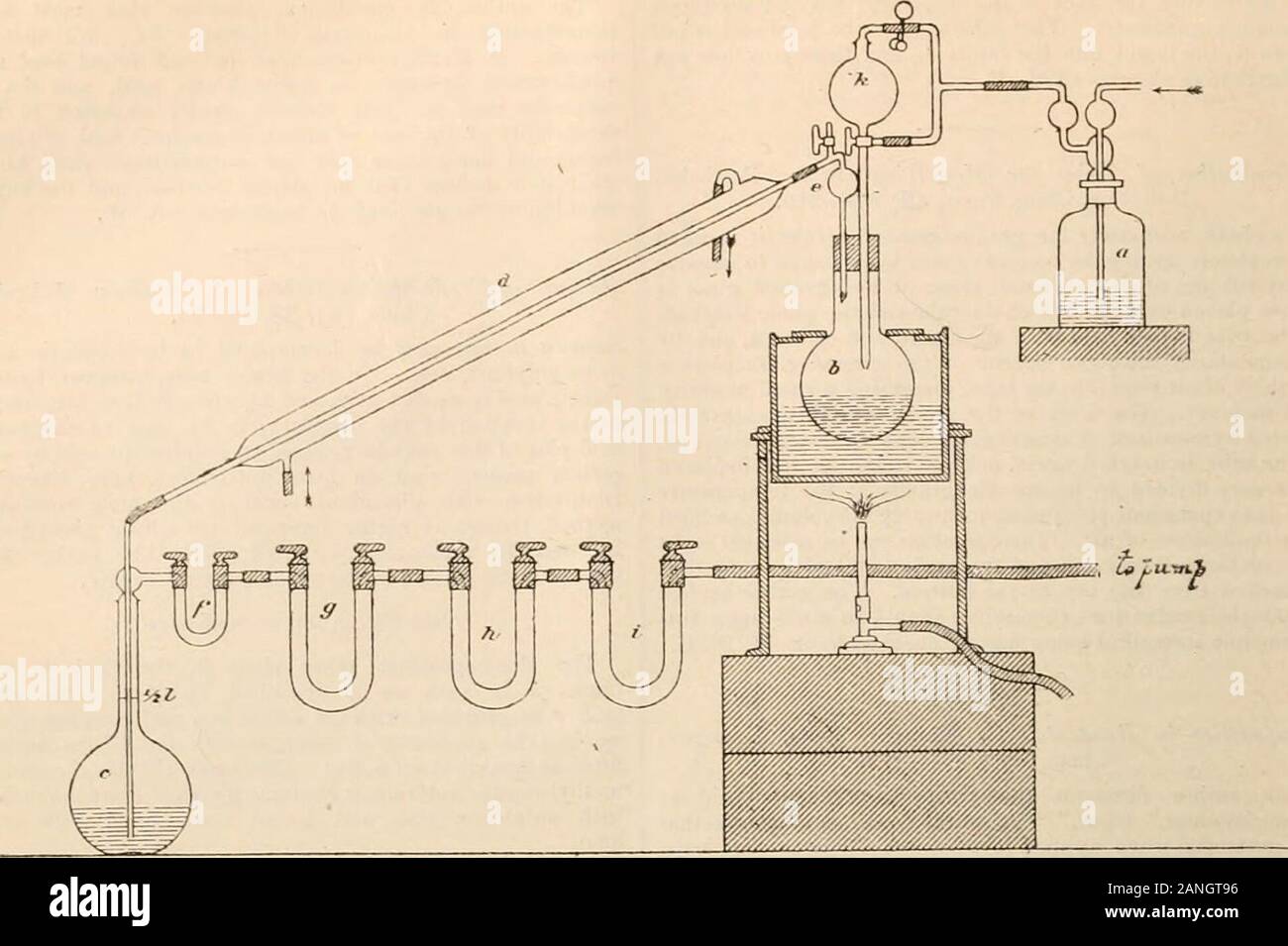
Journal . method. Hedissolves the iron in dilute hydrochloric acid, and passesthe evolved gases by means of ,a stream of hydi-ogenthrough dilute hydrogen peroxide (2-5 per cent.) contain-ing a little caustic soda,

Write the neutralization reaction between Hydrochloric acid HCI and sodium hydroxide NaOH, and write the equation for this process.