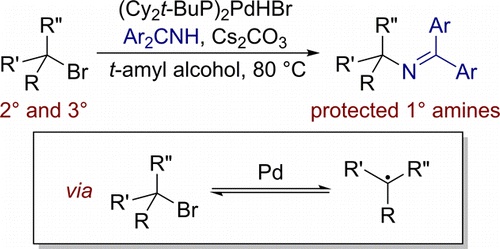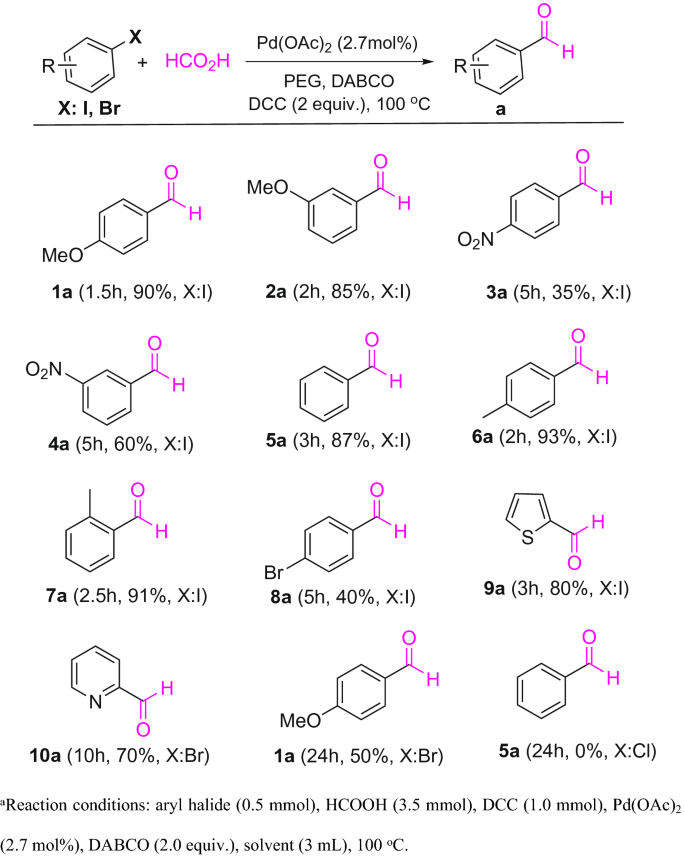
Powerful and Phosphine-Free Palladium-Catalyzed Selective Formylation of Aryl Halides with Formic Acid as CO Source | SpringerLink
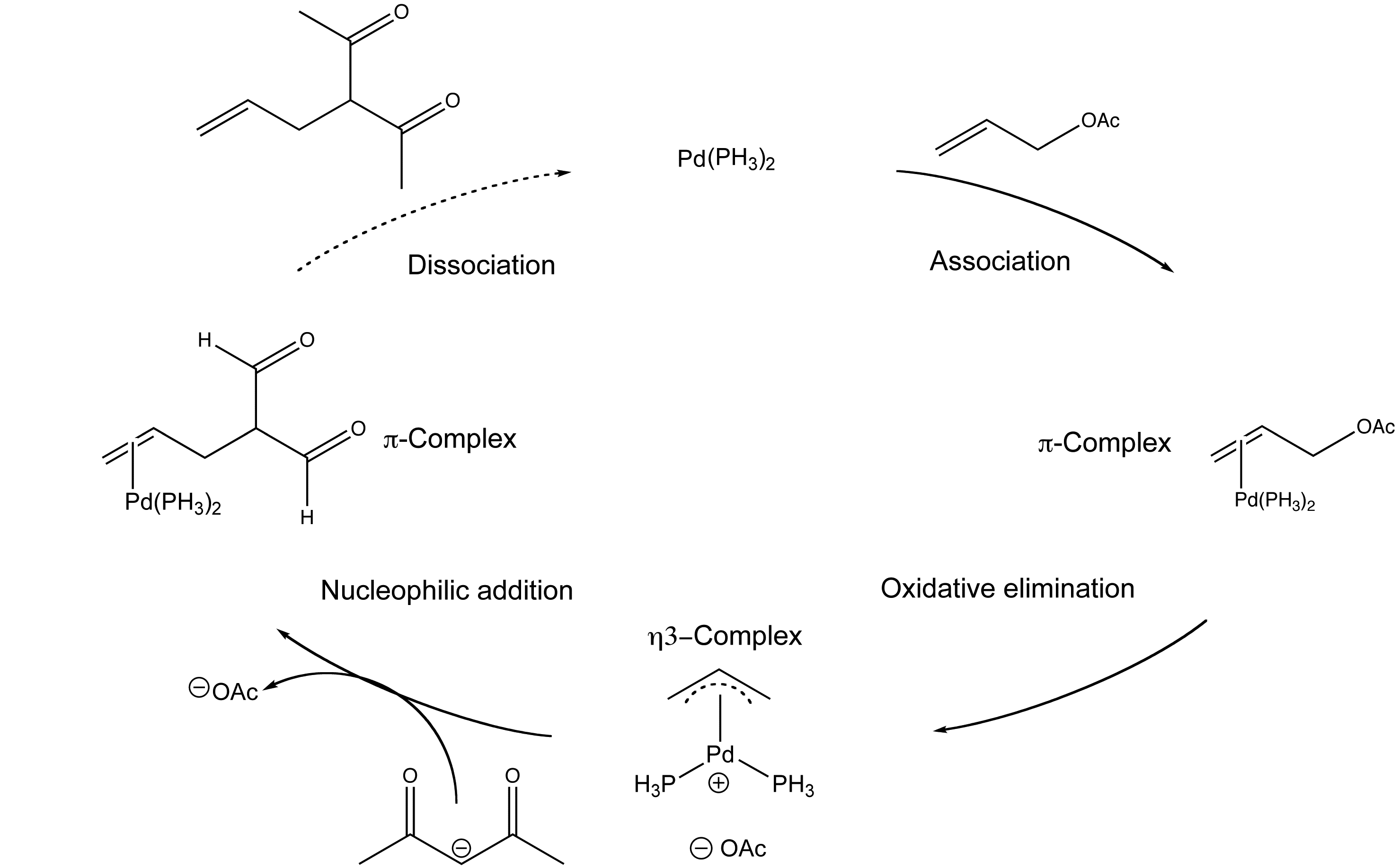
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds

Palladium(II) bromide, Premion , 99.99% (metals basis), Pd 39.5% min, Thermo Scientific Chemicals, Quantity: 2 g | Fisher Scientific

Palladium-Catalyzed Carbohalogenation: Bromide to Iodide Exchange and Domino Processes | Journal of the American Chemical Society
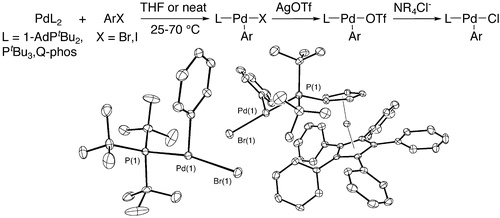
Synthesis, Structure, Theoretical Studies, and Ligand Exchange Reactions of Monomeric, T-Shaped Arylpalladium(II) Halide Complexes with an Additional, Weak Agostic Interaction | The Hartwig Group
![Synthesis of Palladium Benzyl Complexes from the Reaction of PdCl2[P(OPh)3]2 with Benzyl Bromide and Triethylamine: Important Intermediates in Catalytic Carbonylation | Organometallics Synthesis of Palladium Benzyl Complexes from the Reaction of PdCl2[P(OPh)3]2 with Benzyl Bromide and Triethylamine: Important Intermediates in Catalytic Carbonylation | Organometallics](https://pubs.acs.org/cms/10.1021/om010541c/asset/images/om010541c.social.jpeg_v03)
Synthesis of Palladium Benzyl Complexes from the Reaction of PdCl2[P(OPh)3]2 with Benzyl Bromide and Triethylamine: Important Intermediates in Catalytic Carbonylation | Organometallics

TCI Practical Example: One-Pot Palladium-Catalyzed Fluorosulfonylation of an Aryl Bromide Using DABSO and NFSI | TCI AMERICA