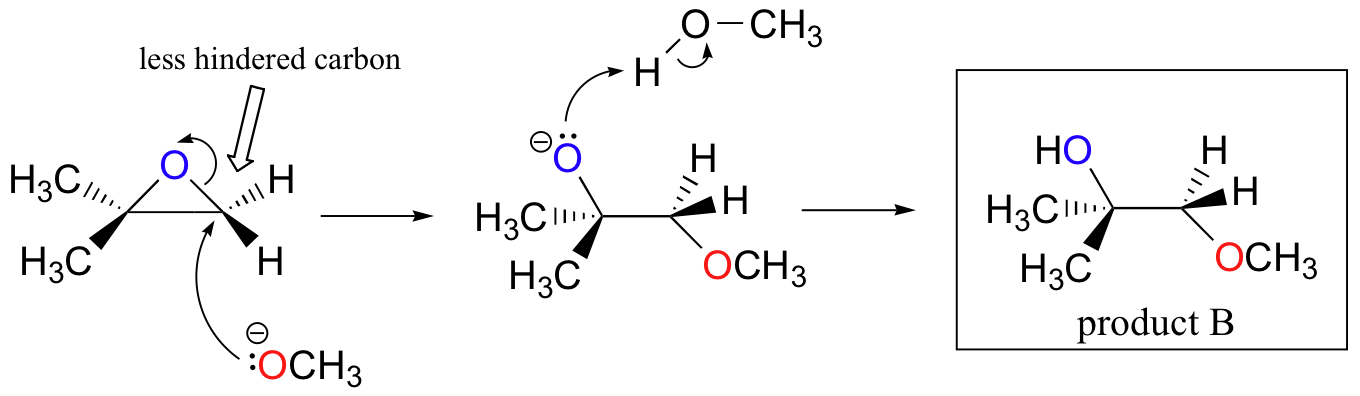
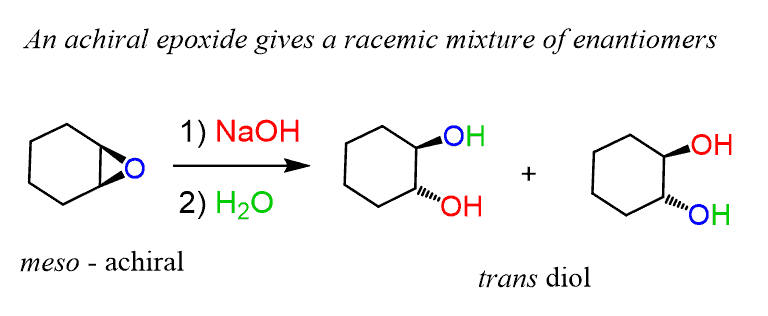
Mechanism and kinetics of epoxide ring-opening with carboxylic acids catalyzed by the corresponding carboxylates - ScienceDirect
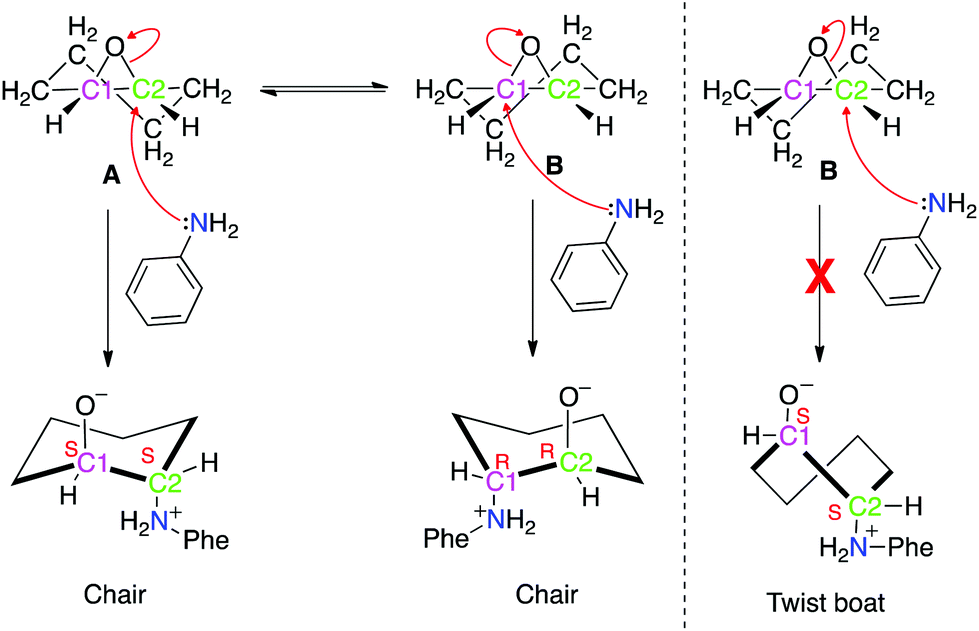
The mechanism of an asymmetric ring-opening reaction of epoxide with amine catalyzed by a metal–organic framework: insights from combined quantum mech ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT04745A
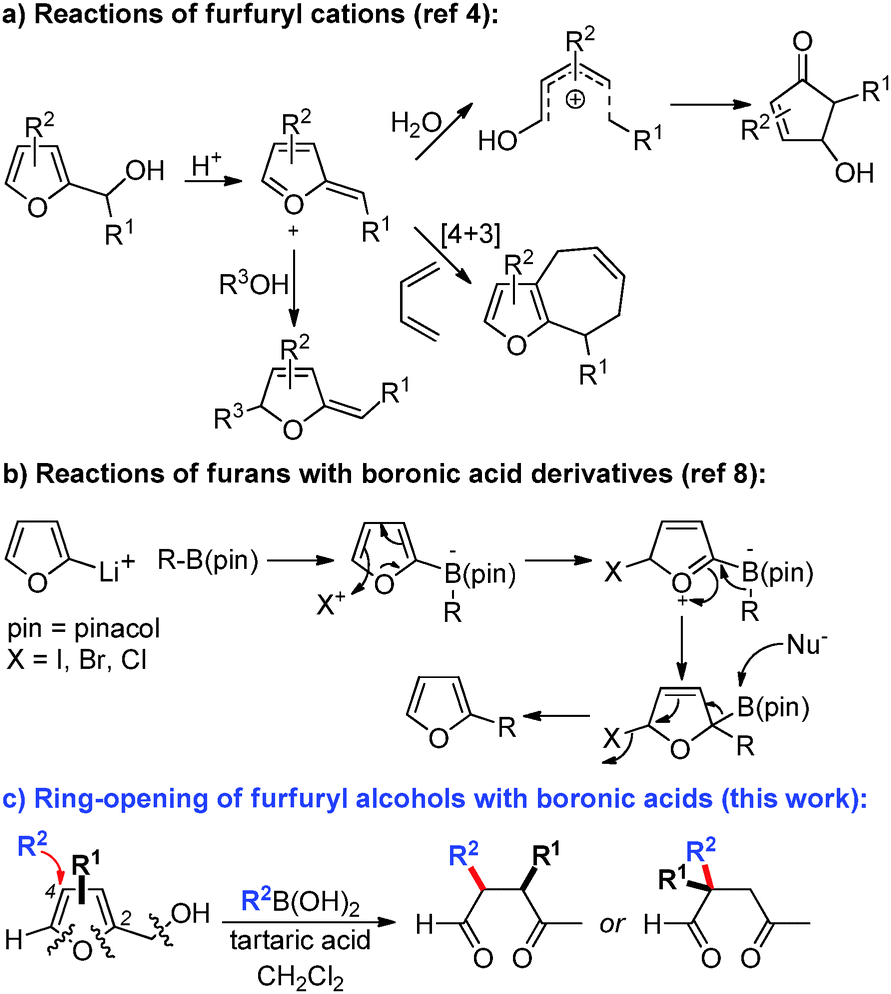
Transition-metal free reactions of boronic acids: cascade addition – ring- opening of furans towards functionalized γ-ketoaldehydes - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC08809G
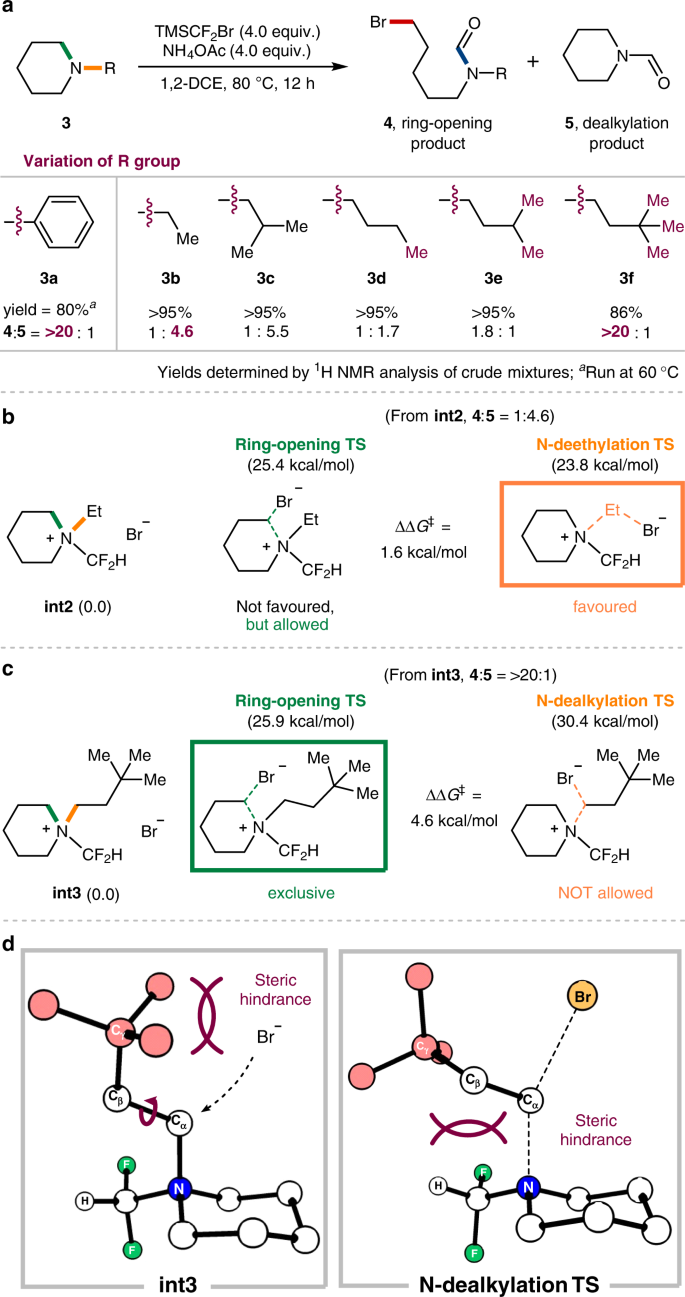
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications

Scheme (3) Mechanism of Ring opening reaction of cellulose-g-Maleic... | Download Scientific Diagram
![Exploring the ring-opening reactions of imidazo[1,5- a ]quinolines for the synthesis of imides under photochemical conditions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01227C Exploring the ring-opening reactions of imidazo[1,5- a ]quinolines for the synthesis of imides under photochemical conditions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01227C](https://pubs.rsc.org/image/article/2019/OB/c9ob01227c/c9ob01227c-s1_hi-res.gif)
Exploring the ring-opening reactions of imidazo[1,5- a ]quinolines for the synthesis of imides under photochemical conditions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01227C

Ultrafast Ring-Opening Reaction of 1,3-Cyclohexadiene: Identification of Nonadiabatic Pathway via Doubly Excited State | Journal of the American Chemical Society

Ring Opening of Epoxides and Aziridines with Sodium Azide using Oxone® in Aqueous Acetonitrile: A Highly Regioselective Azidolysis Reaction

Ring‐Opening of Epoxides in Water - Bonollo - 2011 - European Journal of Organic Chemistry - Wiley Online Library









![Epoxy ring opening reactions with various ring opening reagents [57]. | Download Scientific Diagram Epoxy ring opening reactions with various ring opening reagents [57]. | Download Scientific Diagram](https://www.researchgate.net/publication/330975710/figure/fig1/AS:724209202368513@1549676337569/Epoxy-ring-opening-reactions-with-various-ring-opening-reagents-57_Q640.jpg)