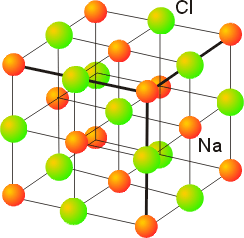
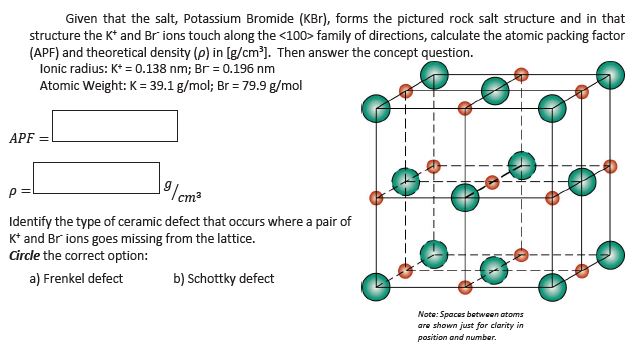
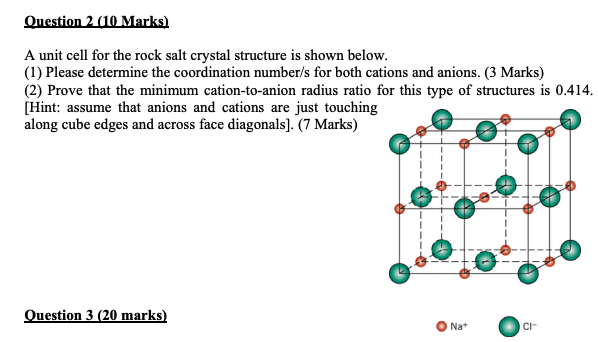
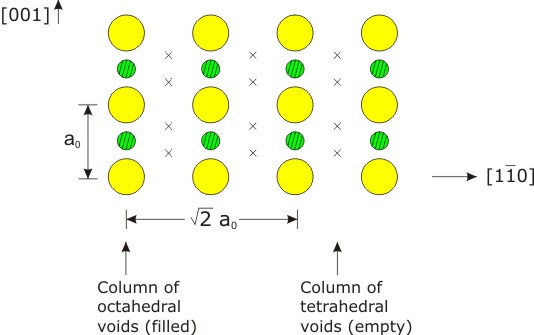
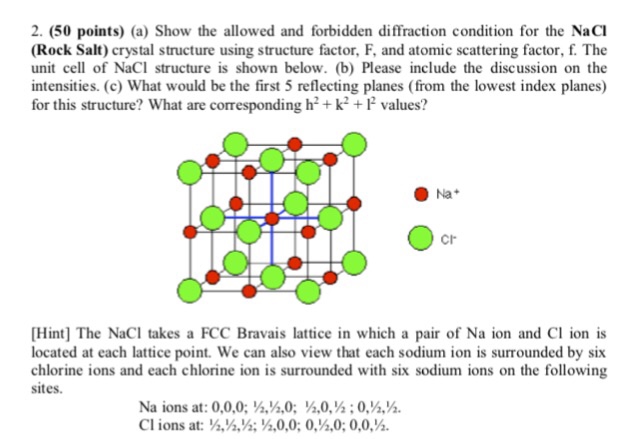
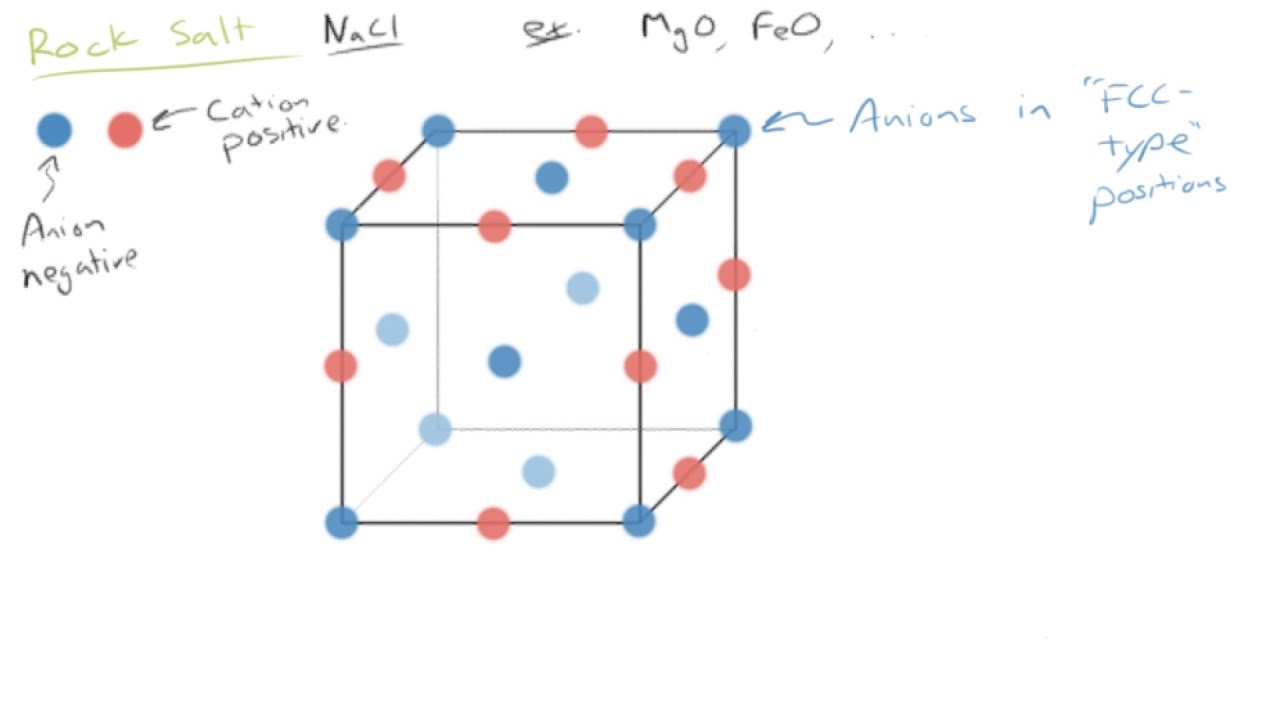
SOLVED:Potassium chloride crystallizes in the rock salt structure. Estimate the density of potassium chloride using the ionic radii provided in Chapter 3.
ionic compounds - Rock salt structure: chloride lattice or sodium lattice? - Chemistry Stack Exchange
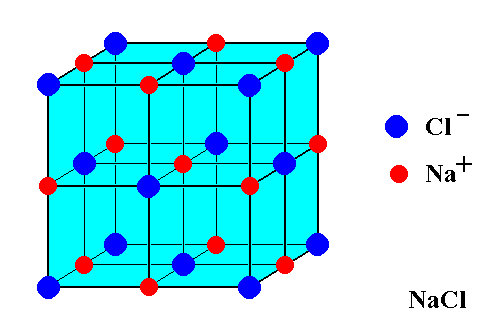
The edge length of the rock salt type unit cell is 508 pm. If the radius of the cation is 110 pm, the radius of the anion assuming NaCl type structure is:A.
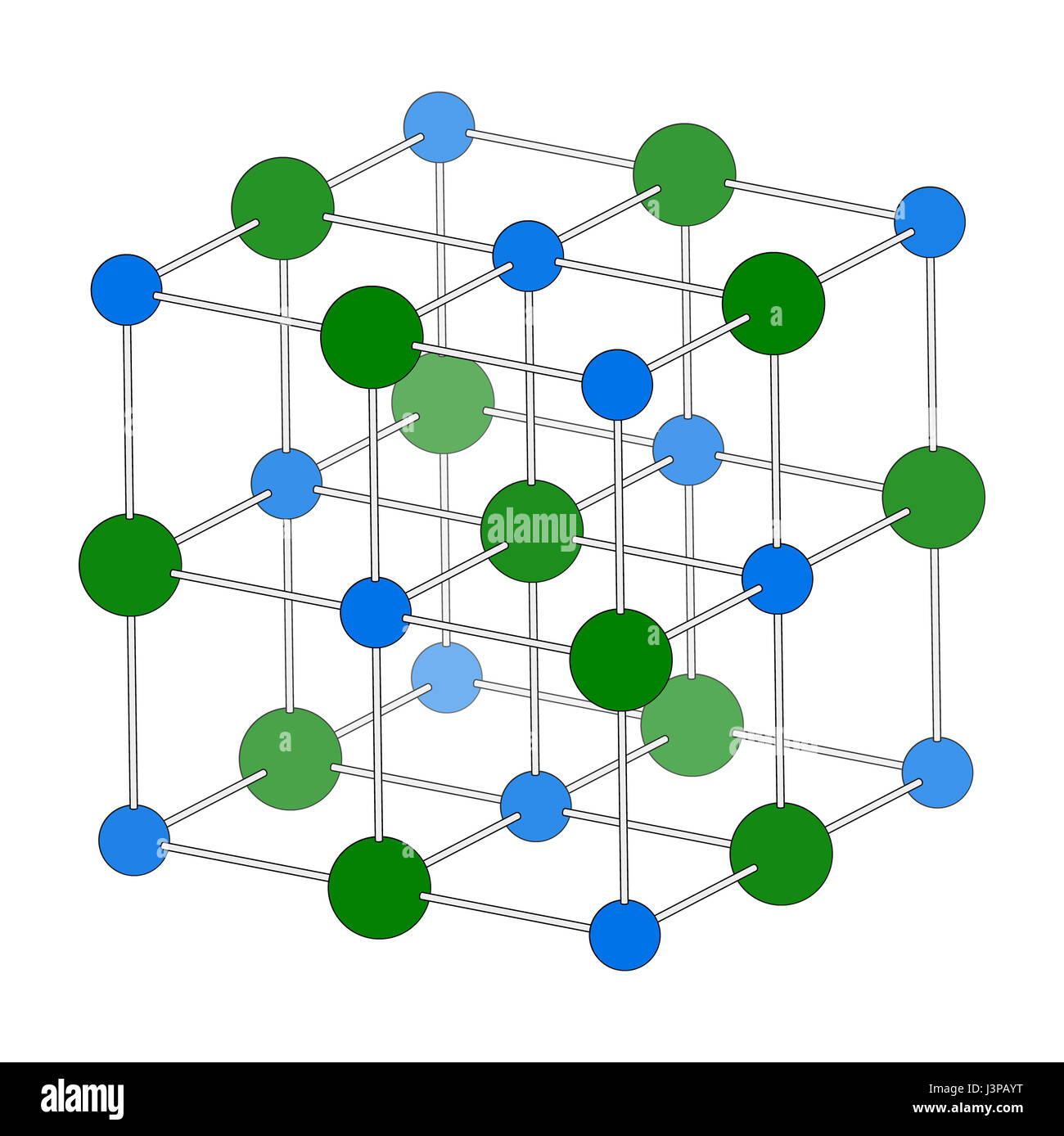
Sodium chloride (rock salt, halite, table salt), crystal structure. Atoms shown as color-coded spheres (Na, blue; Cl, green). Unit cell Stock Photo - Alamy

The common rocksalt crystal structure of the RENs. The large spheres... | Download Scientific Diagram

Sodium chloride (rock salt, halite, table salt), crystal structure, Canvas Print | Barewalls Posters & Prints | bwc27518114

From Trigonal to Cubic LiVO2: A High-Energy Phase Transition toward Disordered Rock Salt Materials | The Journal of Physical Chemistry C

Phase transition in SiC from zinc-blende to rock-salt structure and implications for carbon-rich extrasolar planets

Unveiling composition/crystal structure-dependent electrochemical behaviors via experiments and first-principles calculations: rock-salt NiCoO2 vs. spinel Ni1.5Co1.5O4 - ScienceDirect

Charged rock-salt structure where two elements of the neutral rock-salt... | Download Scientific Diagram

Disordered Rock Salt And Transition Metal Anodes — Engineering The Batteries Of The Future - CleanTechnica