3. 'van 't Hoff plot for toluene' demonstrating curvilinear behavior... | Download Scientific Diagram
![Graph between log k and 1/T [k is rate constant (s^-1) and T the temperature (K)] is a straight line with OX = 5, theta = tan^-1 (1/2.303). Hence - Ea will be : Graph between log k and 1/T [k is rate constant (s^-1) and T the temperature (K)] is a straight line with OX = 5, theta = tan^-1 (1/2.303). Hence - Ea will be :](https://dwes9vv9u0550.cloudfront.net/images/2653450/b830d43b-5f91-4a2b-a361-907220a672f5.jpg)
Graph between log k and 1/T [k is rate constant (s^-1) and T the temperature (K)] is a straight line with OX = 5, theta = tan^-1 (1/2.303). Hence - Ea will be :

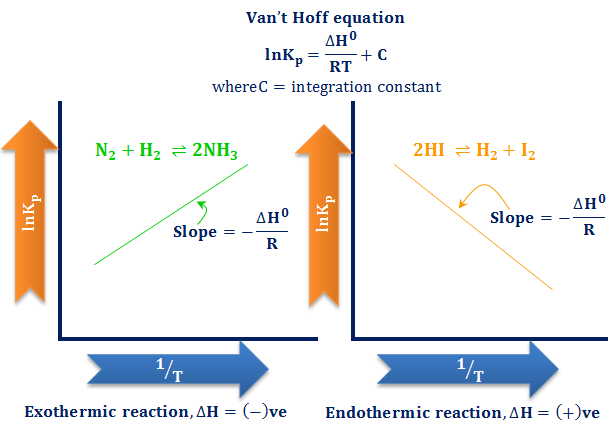
Lesson 19-van't hoff equation - Vant Hoff Equation The vant Hoff equation can be used to estimate Kc or Kp at a given temperature, provided that Kc or | Course Hero