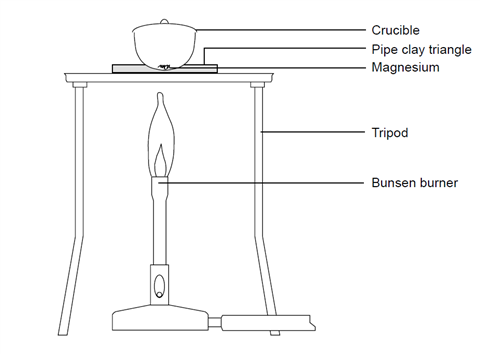
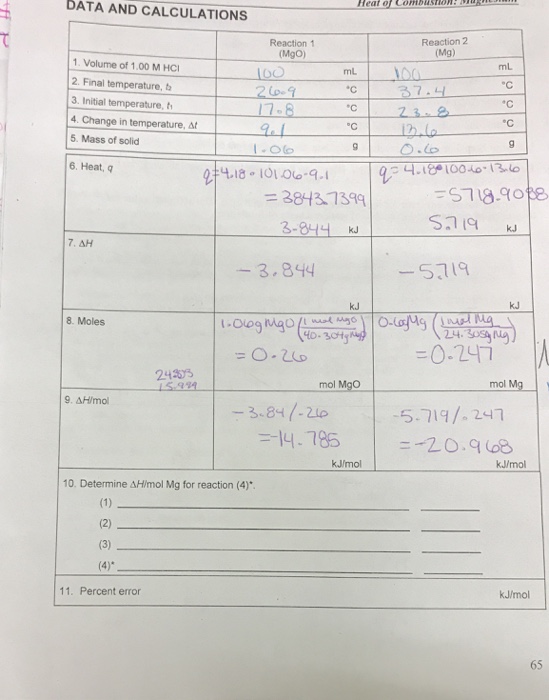
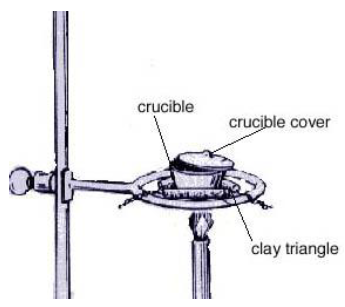
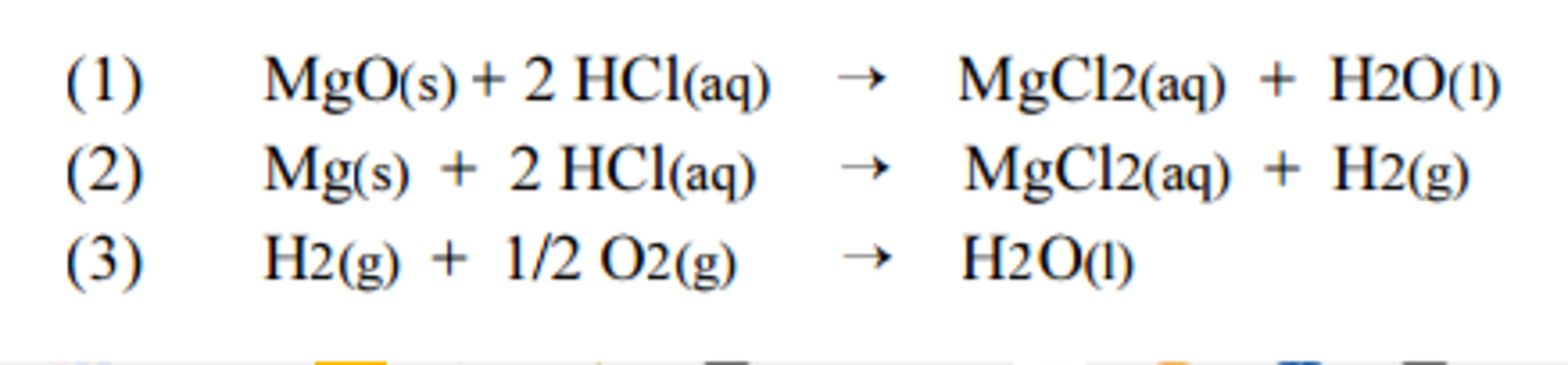Hesss Law Lab, use Hesss law to find the enthalpy change of combustion of magnesium which is a highly exothermic reaction. - International Baccalaureate Chemistry - Marked by Teachers.com
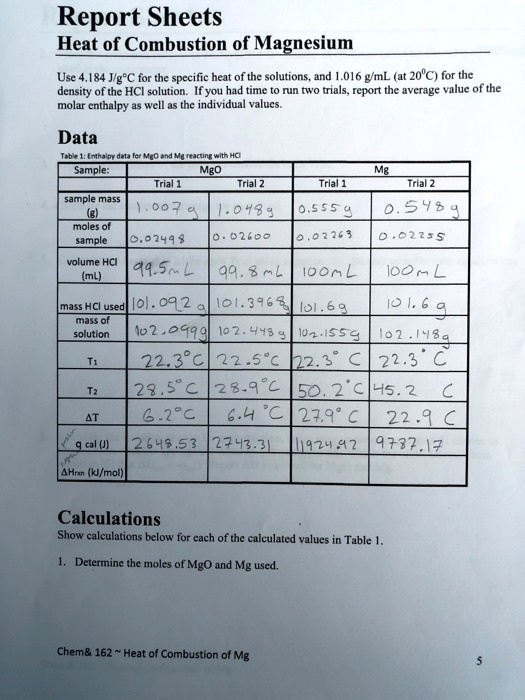
SOLVED: Report Sheets Heat of Combustion of Magnesium Use 184 Jlg"C for the specific heat of the solutions, and 1.016 gmL (at 20"C) for the density of the HCI solution. Ifyou had

Grade 12 Chemistry Lab: Hess's Law and the Enthalpy of Combustion of Magnesium | Chemistry (University) - Grade 12 OSSD | Thinkswap