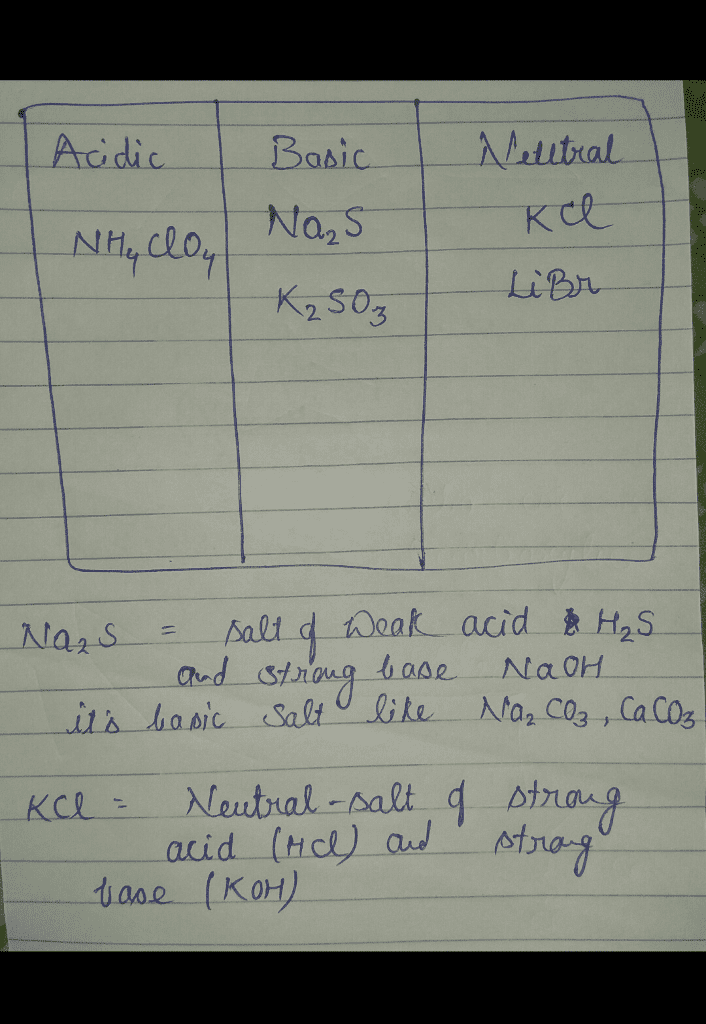
SOLVED: Are each ofthe following an acidic, basic, or neutral salt? A. Basic salt B. Neutral salt C. Acidic salt select KHzPO4 select 2. AICl3 select 3.NHACzH3O2 select 4. CsH6NCI

Acidic & Basic Salt Solutions & Examples | What are Acidic & Basic Salts? - Video & Lesson Transcript | Study.com

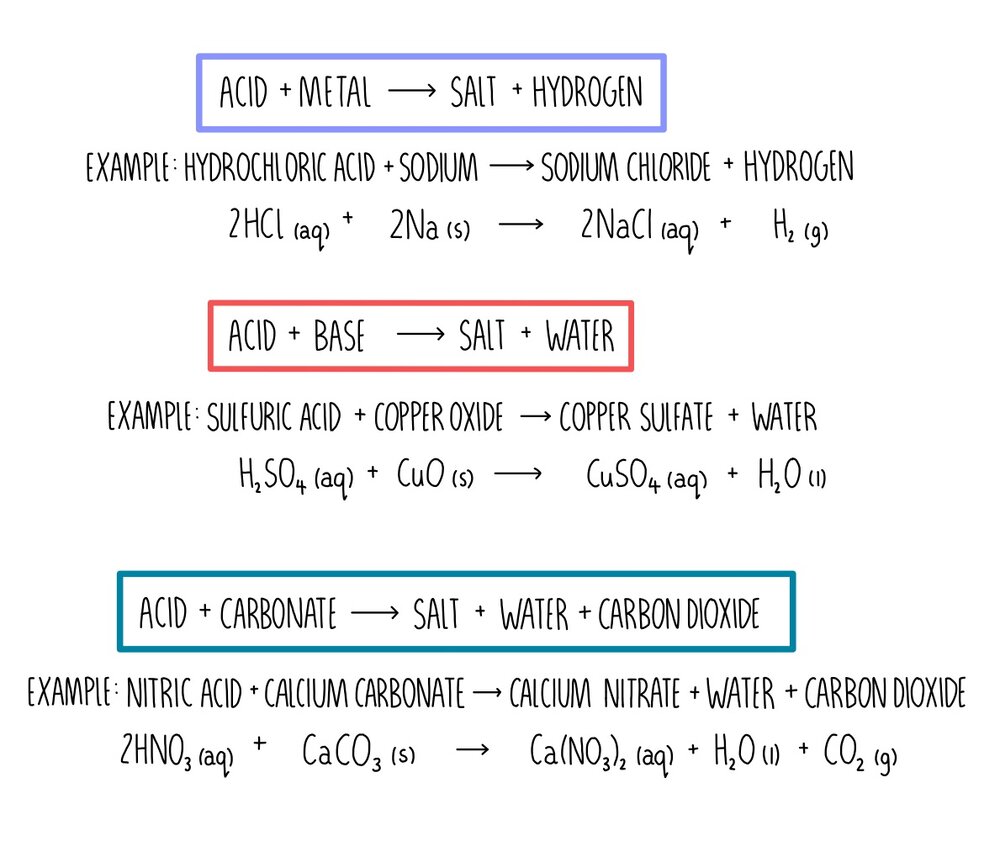
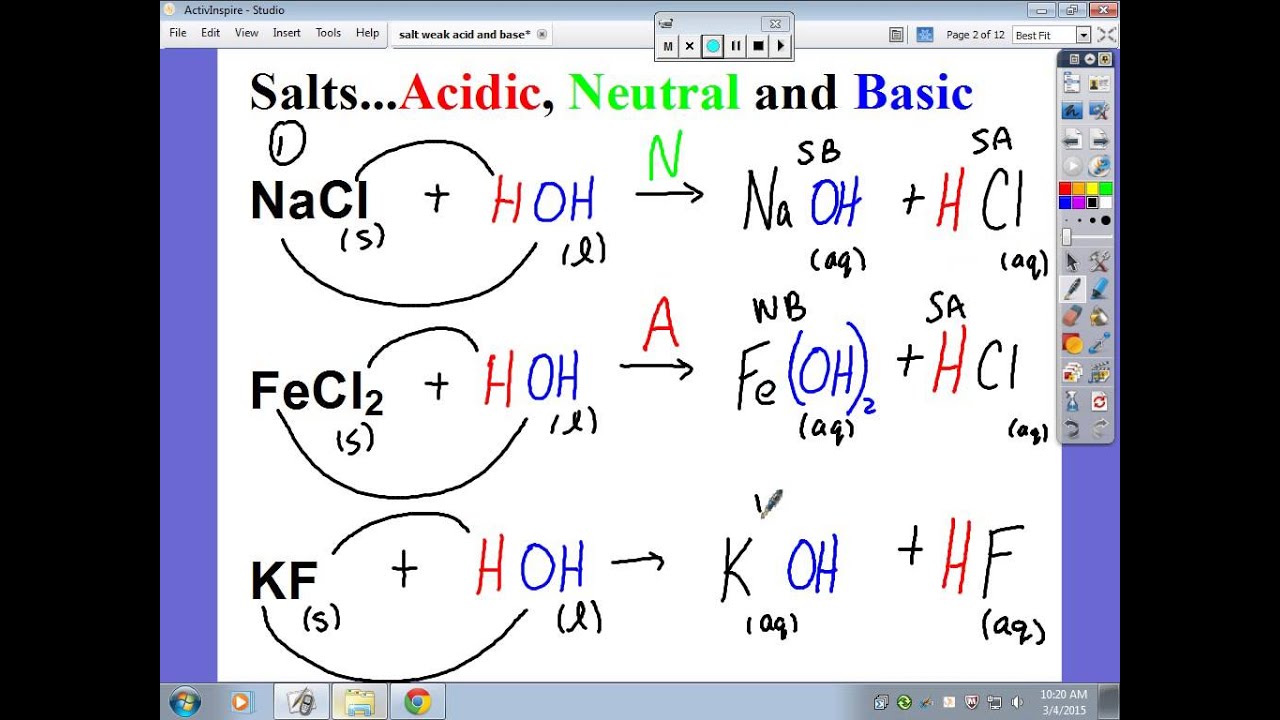
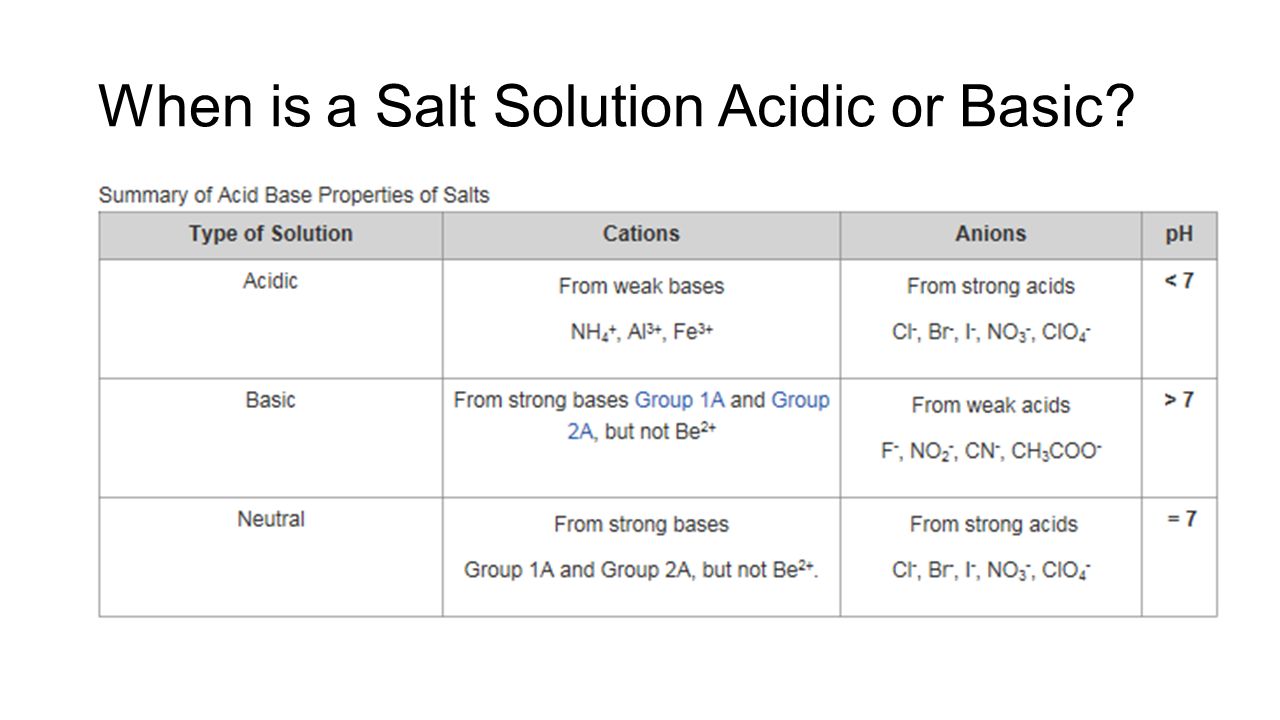
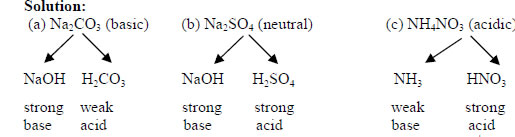
PH of Salts When acids are neutralized by bases, water and a salt is formed. The salt solution can be acidic, basic, or neutral depending on the acid. - ppt download
Comparison of the effects of conceptual change texts implemented after and before instruction on secondary school students' understanding of acid-base concepts