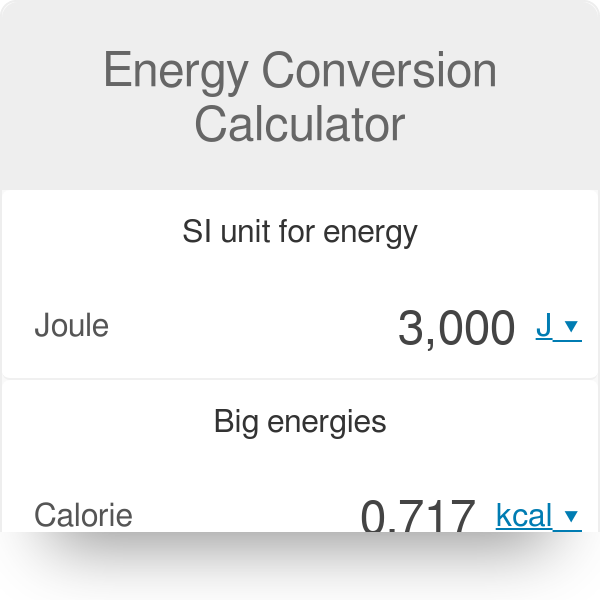
The heat Q that must be supplied or removed to change the temperature of a substance of mass m by an amount ∆T is: Q = cm∆T where c is the specific. -
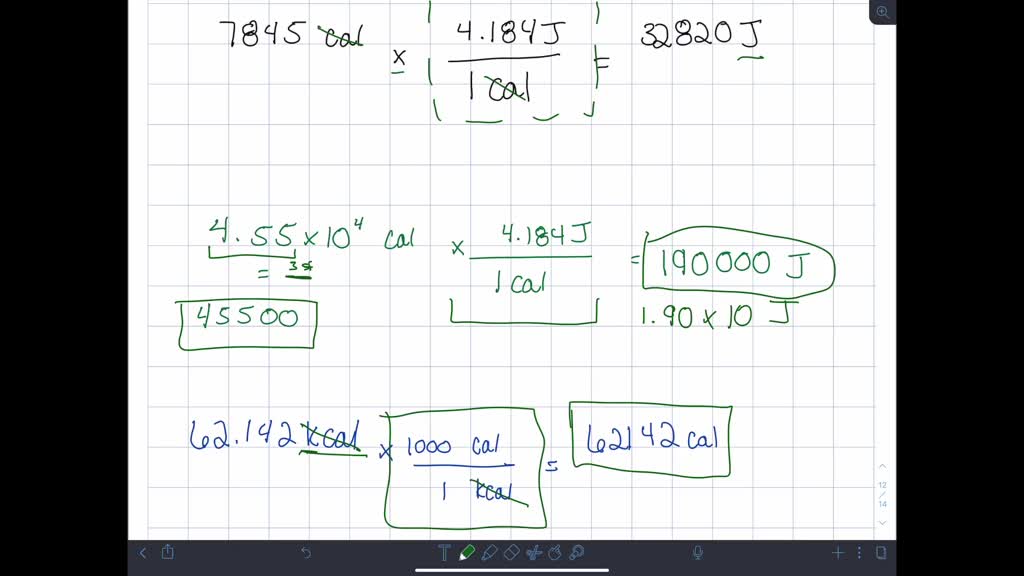
SOLVED:Convert the following numbers of calories or kilocalories into joules or kilojoules. a. 7845 cal b. 4.55 ×10^4 cal c. 62.142 kcal d. 43,024 cal

5.03 Conversion of kilojoules to calories | Year 11 Maths | WACE 11 Essential Mathematics - 2020 Edition | Mathspace

Chapter 6. Calorie One calorie expresses the quantity of heat necessary to raise the temperature of 1 g of water by 1° Celsius. Kilocalorie (kCal) - ppt download

SOLVED:Perform the indicated conversions. a. 91.74 kcal into calories b. 1.781 kJ into calories c. 4.318 ×10^3 J into kilocalories d. 9.173 ×10^4 cal into kilojoules

WSheatconversions (1).doc - NAME: DATE: 1. Convert 23 calories into kcal. 2. Convert 7.9 kcal into calories. 3. Convert 234 J into kJ. 4. Convert 21.3 | Course Hero