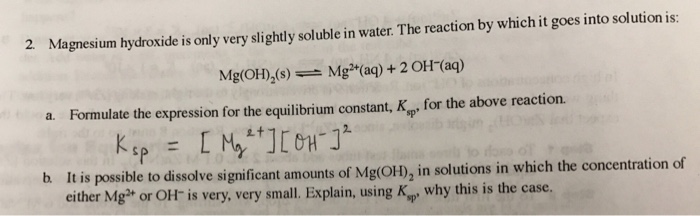Arrange the following metal in the increasing order of their reactivity towards water: Zinc, Iron, Magnesium, Sodium.

Question Video: Ordering the Reactions of Magnesium Metal with Varying Concentrations of Nitric Acid | Nagwa

SOLVED: antacid; Milk of Magnesia suspension of magnesium hydroxide,Mg(OH)z: In water, The popular. the reaction shown in Equation Mg(OH)z undergoes anuiniodsl-ied Mg(OH), (s, white) Mg"* (aq) 2 OH (4q1 (Eq, 9 What

What happens when Magnesium oxide is dissolved in water? Write a word equation for this process. Name - Brainly.in