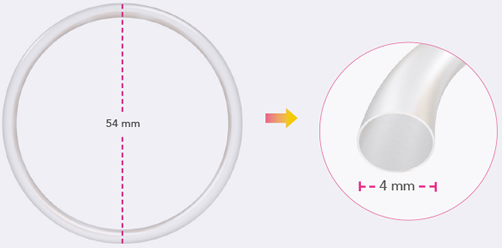
Pharmacokinetic bioequivalence, safety and acceptability of Ornibel®, a new polymer composition contraceptive vaginal ring (eto
Non-interventional retrospective study to evaluate menstrual bleeding pro les, tolerability and quality of life of women using t
Pharmacokinetic bioequivalence, safety and acceptability of Ornibel®, a new polymer composition contraceptive vaginal ring (eto

PDF) Pharmacokinetic bioequivalence, safety and acceptability of Ornibel ® , a new polymer composition contraceptive vaginal ring (etonogestrel/ethinylestradiol 11.00/3.474 mg) compared with Nuvaring ® (etonogestrel/ethinylestradiol 11.7/2.7 mg)