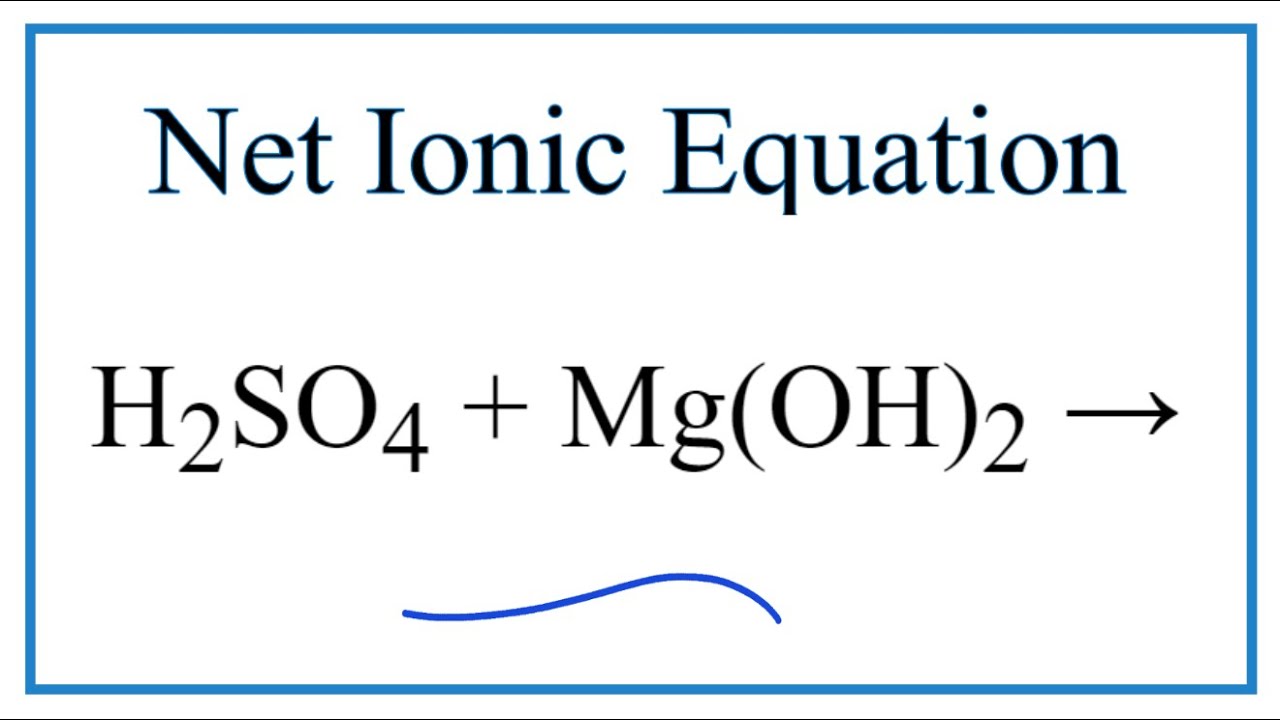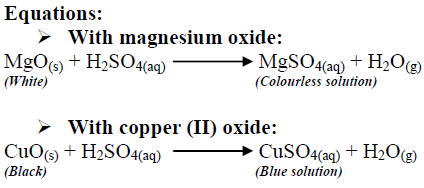What affects the reaction rate between magnesium and sulphuric acid? - GCSE Science - Marked by Teachers.com

investigate reactions between dilute hydrochloric and sulfuric acids and metals (e.g. magnesium, zinc and iron) Diagram | Quizlet

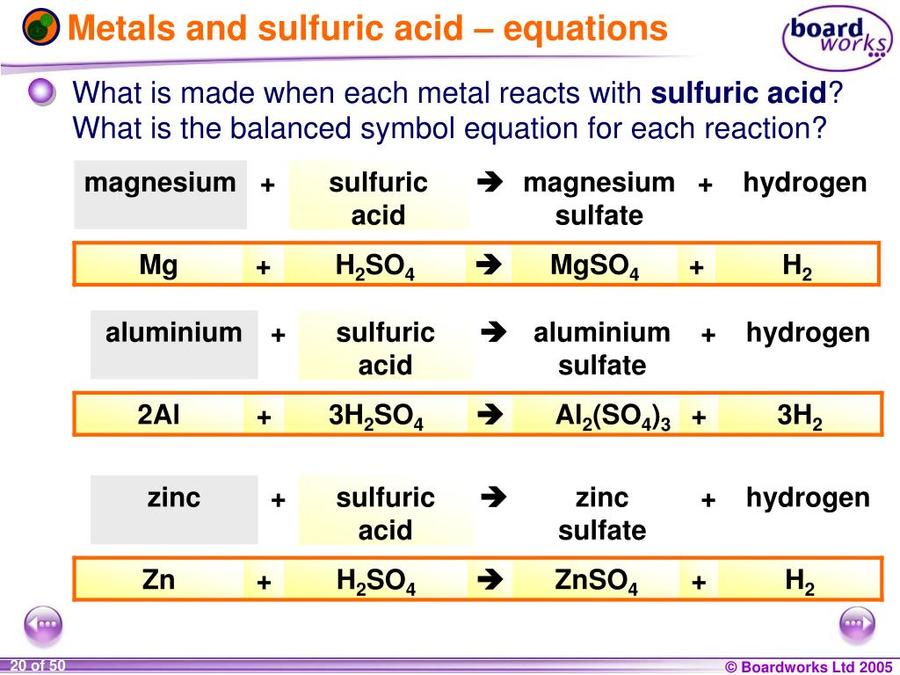
Write word equations and then balanced equations for the reaction taking place when:(a) Dilute sulphuric acid reacts with zinc granules.(b) Dilute hydrochloric acid reacts with magnesium ribbon.(c) Dilute sulphuric acid reacts with

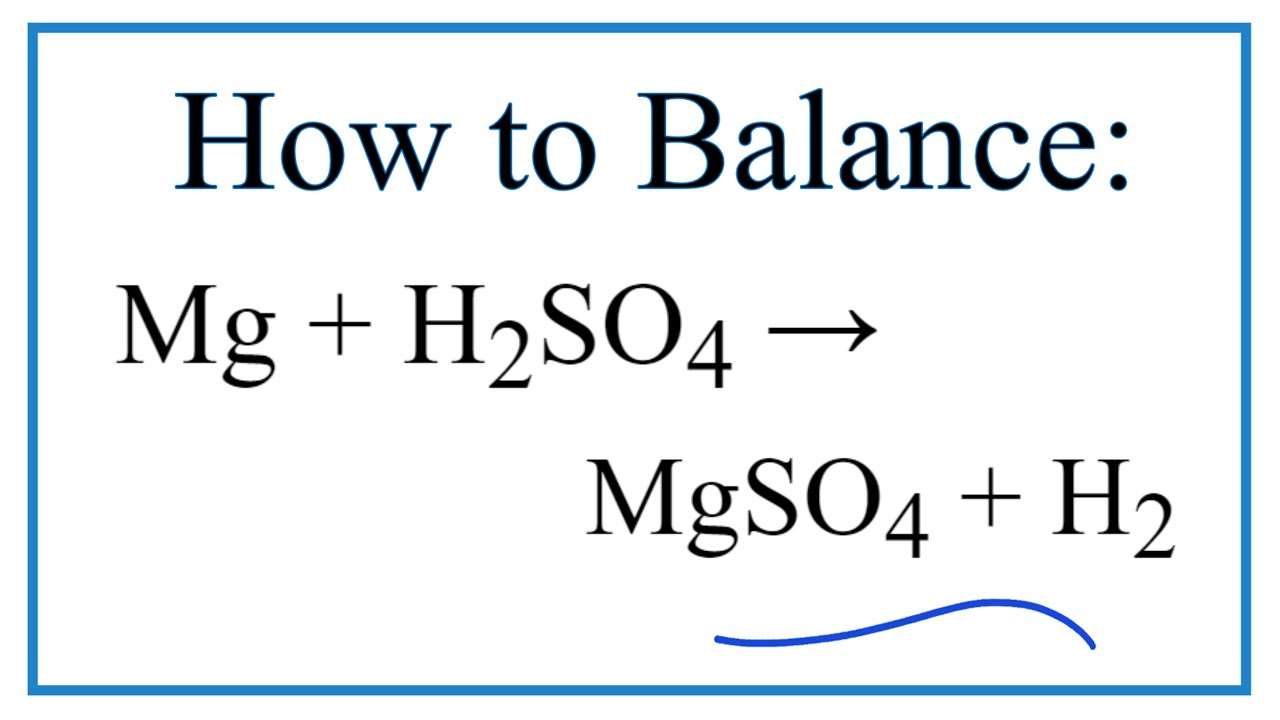
The equation shows the reaction between magnesium and sulphuric acid: Mg + H2SO4 ⟶ MgSO4 + H2 In this reaction, calculate the mass of magnesium sulphate formed when 6g of magnesium reacts

Investigate reactions between dilute hydrochloric and sulphuric acids and metals (eg. Magnesium, Zinc and Iron) Diagram | Quizlet
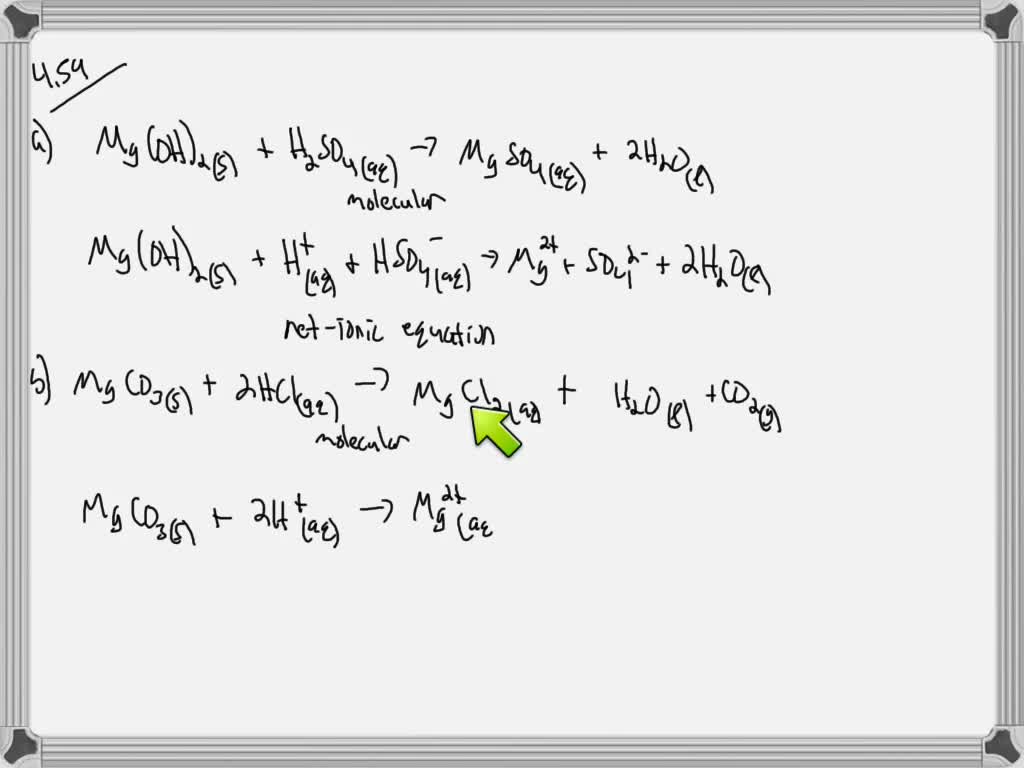
SOLVED:Write a balanced molecular equation and a net ionic equation for the following reactions: a. Solid magnesium hydroxide reacts with a solution of sulfuric acid. b. Solid magnesium carbonate reacts with a
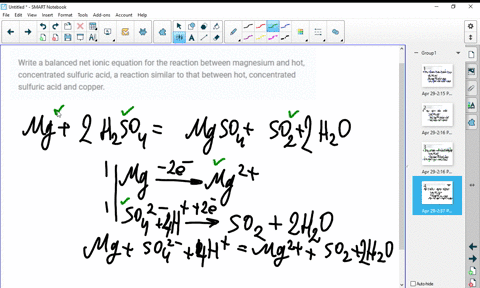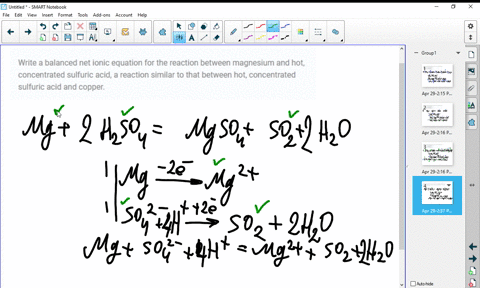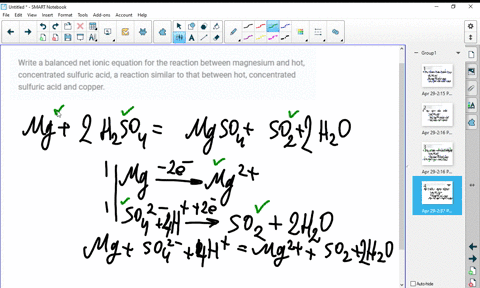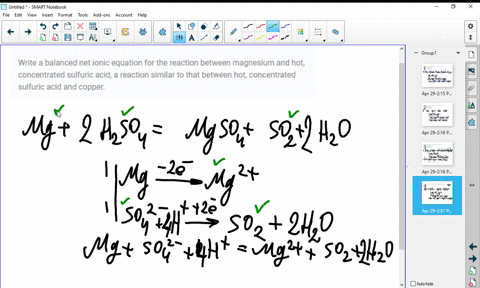
SOLVED:Write a balanced net ionic equation for the reaction between magnesium and hot, concentrated sulfuric acid, a reaction similar to that between hot, concentrated sulfuric acid and copper.

Practical: Investigate Metals Reacting with Acids (2.4.6) | Edexcel IGCSE Chemistry Revision Notes 2019 | Save My Exams