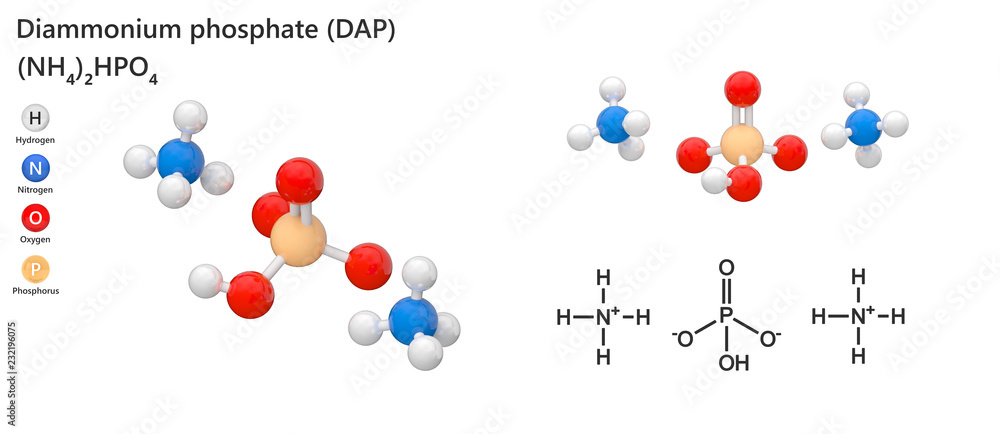
Diammonium phosphate (DAP) (chemical formula (NH4)2HPO4) is one of a series of water-soluble ammonium phosphate salts. 3d illustration. The molecule is represented in different structures. Stock Illustration | Adobe Stock

Disproportionation of Pharmaceutical Salts: pHmax and Phase-Solubility/pH Variance | Molecular Pharmaceutics
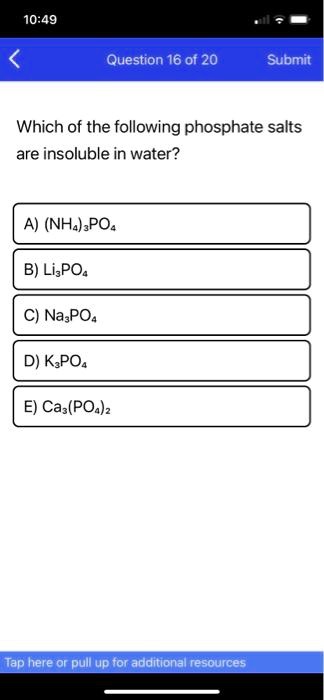
SOLVED: 10.49 Question 16 of 20 Submit Which of the following phosphate salts are insoluble in water? A) (NHa);POa B) LiPOa NasPOa D) K;POa E) Ca,(POa)z ap hereror pull up tor additicnal

SOLVED: Question 10 Status: Not yet answered Points possible: Identify each category of substance as soluble or insoluble in water: Salts of Group elements Choose . Most silver salts Choose . Most
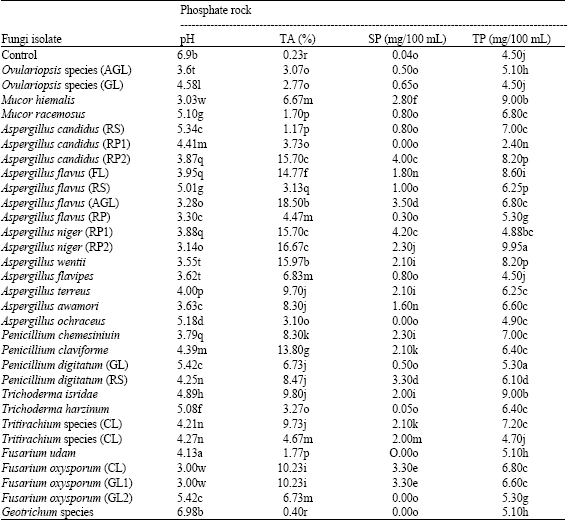
Solubilization of Insoluble Phosphate by Organic Acid-Producing Fungi Isolated from Nigerian Soil - SciAlert Responsive Version

Soluble phosphate salts as setting aids for premixed calcium phosphate bone cement pastes - ScienceDirect

Solubility of Calcium Phosphate in Concentrated Dairy Effluent Brines | Journal of Agricultural and Food Chemistry

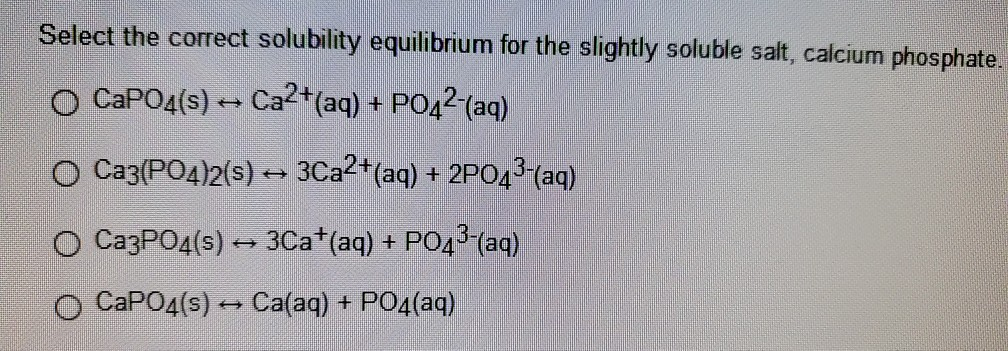





![PDF] EFFECTS OF SOLUBLE SALTS ON INSOLUBLE PHOSPHATES | Semantic Scholar PDF] EFFECTS OF SOLUBLE SALTS ON INSOLUBLE PHOSPHATES | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8a4cd892c77a0bd7c7baf3aa828a051811f98784/23-Table2-1.png)








