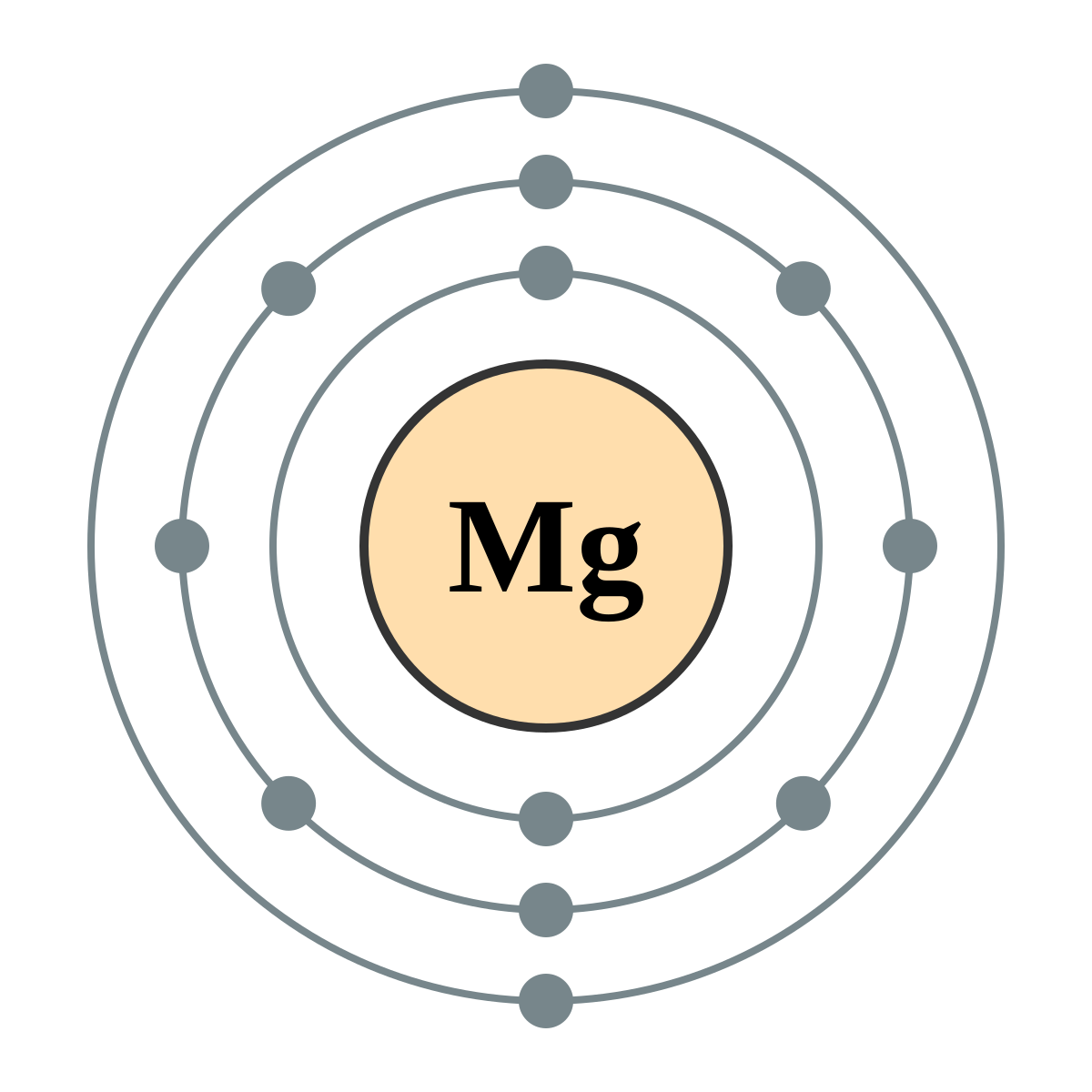
Mg Magnesium Element Information: Facts, Properties, Trends, Uses and comparison - Periodic Table of the Elements | SchoolMyKids

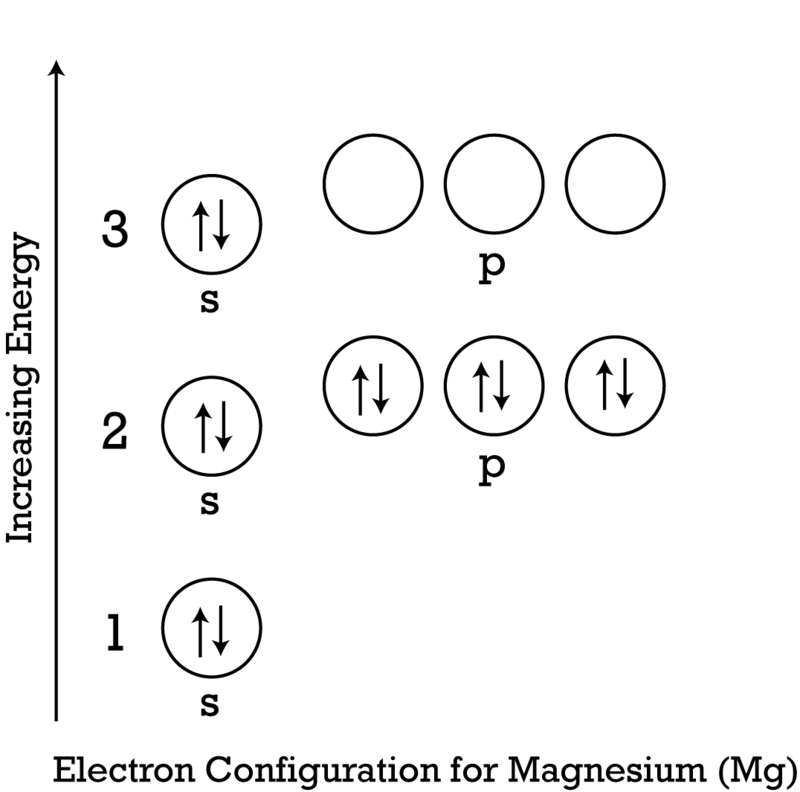
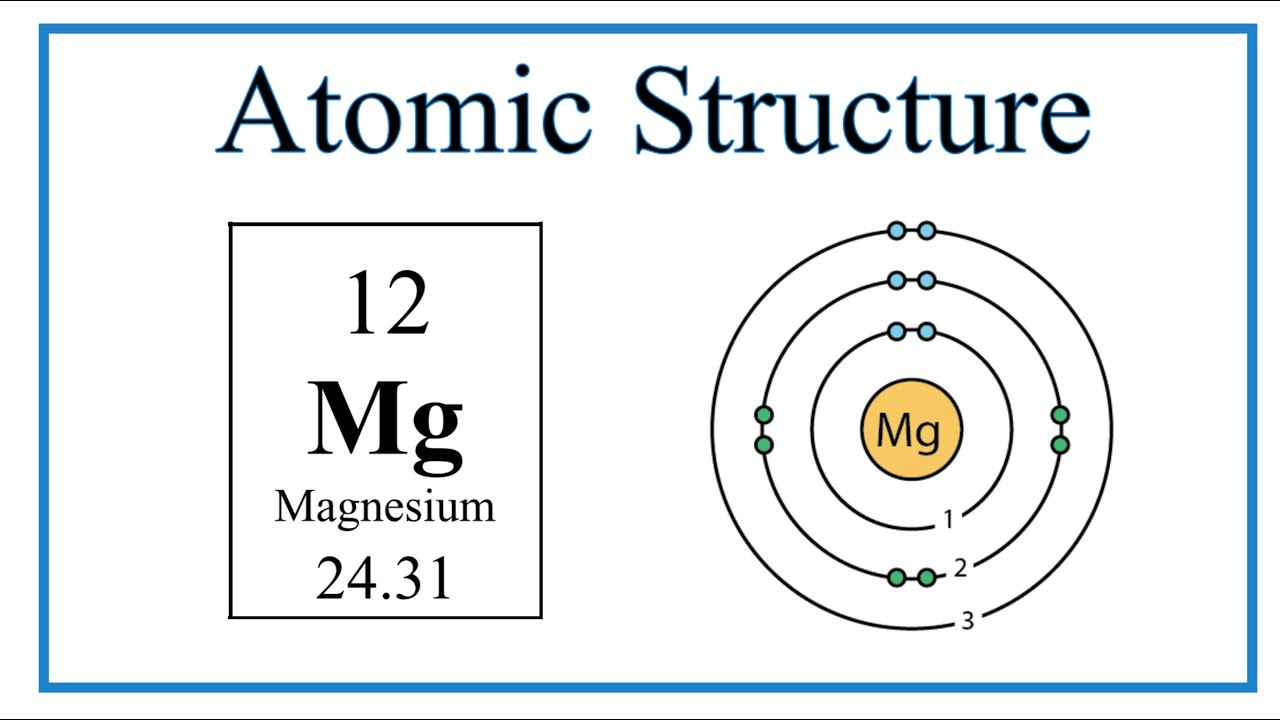
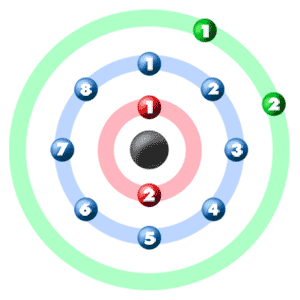
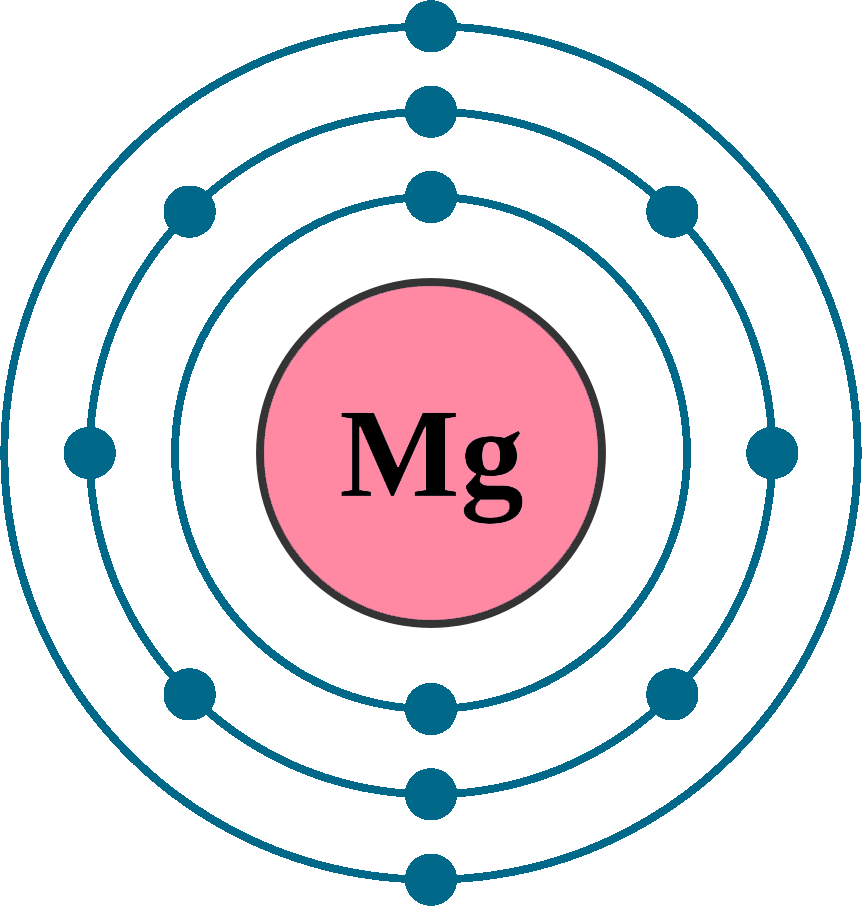
Draw a model of the atom 2512Mg. How many valence electrons does it have? What is the group number of magnesium? | Homework.Study.com
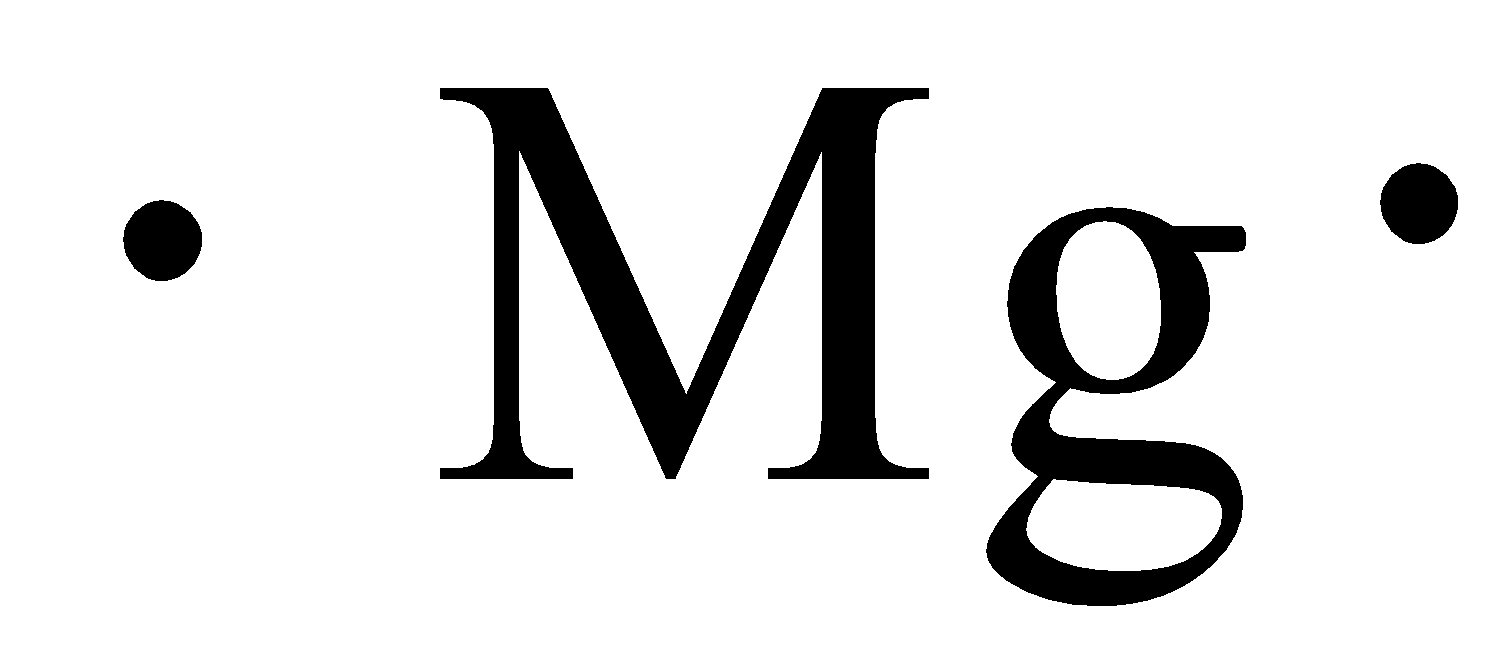
Write the electron dot structure for magnesium and chlorine. Show the formation of magnesium chloride by the transfer of electrons. What are the ions present in this compound?
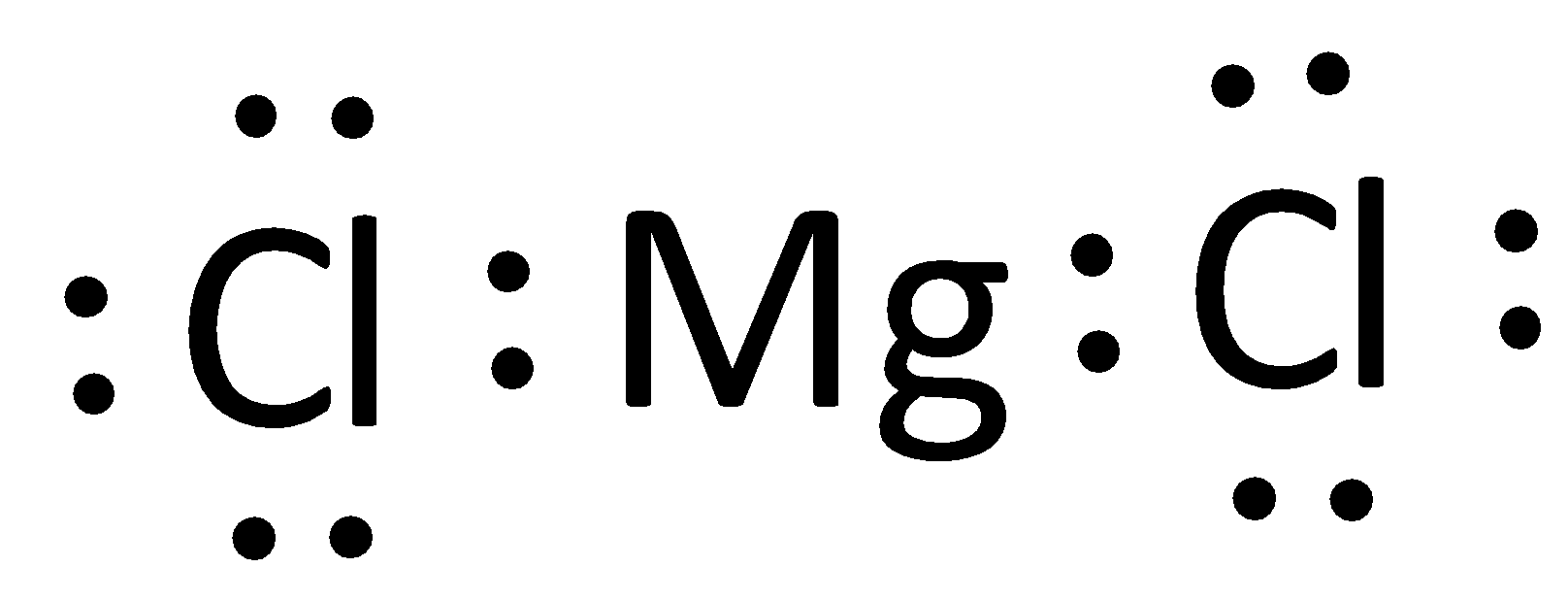
Lewis structure of ${\\text{MgC}}{{\\text{l}}_{\\text{2}}}$ and ${\\text{A}}{{\\text{l}}_{\\text{2}}}{{\\text{O}}_{\\text{3}}}$are:A.\n \n \n \n \n B.\n \n \n \n \n C.Both A and BD.None of the above

Magnesium has 2 valence electrons, and oxygen has 6 valence electrons, which type of bonding is likely to occur between a magnesium atom and an oxygen atom? | Socratic